The Bad Supplements List: Failed and Prohibited Products and Brands
Last revised: February 25, 2026
ABOUT THE BAD SUPPLEMENTS LIST:
The Bad Supplements List documents and reviews clear and reliable evidence, using publicly available information on testing and enforcement activities on non-compliant U.S. dietary supplement brands and products. The List focuses on recent reports of firms, brands and products which may still be present in the market.
Source information on this page is not confidential, and is freely available for public view online. Information may be provided by government, public or private entities with permission.
Bad Supplements is a working document, and will be continuously updated.
HOW TO VIEW:
1. Search by product or company name using Find (Ctrl-F) or the search box at the bottom of the page.
2. Scroll down through this page to the following sections
- NOW Foods Testing of Dietary Supplements
- FDA Warning Letters
- Flagrant DOJ Actions, Court Orders and Injunctions
3. Visit the List of Prohibited Supplements on Amazon.com
We recommend all supplement consumers to verify all brand and product information with the brand owner, and to research enforcement action on all brands and products before consuming. Recommended search terms typically include the regulatory agency (e.g. FDA, FTC) and the brand or product name.
Some sources cited may require access or subscription fees from the publisher.
DISCLOSURES:
NaturPro Scientific has no financial interest in any dietary supplement brand or product. We do not accept payment for removal from or inclusion on this List. Bad Supplements is made possible with support from responsible firms in the dietary supplement industry. Please contact us to learn how to support the Bad Supplements List.
Information posted by NaturPro Scientific LLC on the Bad Supplements List or elsewhere are based on an expert review based on reasonable interpretation of regulatory requirements and mainly public information. Private information may be posted, only with permission from the owner. We do not disclose any information which is prohibited from public view, or considered confidential or proprietary.Although we take responsibility for reviewing information herein to reliable sources, we are not responsible for the accuracy of information provided by any third parties. Information on this page may be updated or changed without notice.
NOW Tests St. John’s Wort Supplements Sold on Amazon (October 29, 2025)
Continuing our mission to promote transparency and higher standards in the supplement industry, NOW recently tested 22 St. John’s Wort products purchased from Amazon in August 2025. Results revealed that all except for one (NOW) product failed potency testing, and several contained synthetic dyes used to make low-quality or adulterated products appear authentic.
We purchased two bottles of each St. John’s Wort supplement (including NOW product) to conduct internal analyses, and we partnered with Alkemist Labs, an ISO 17025-accredited independent laboratory specializing in botanical identification and testing, to ensure accuracy and transparency.
Why Test St. John’s Wort?
St. John’s Wort (Hypericum perforatum) is a popular herb used to support mood balance and emotional well-being. The key active compounds, hypericin and pseudohypericin, are responsible for its beneficial properties and are typically standardized to 0.3%. Ensuring supplements contain genuine, properly standardized extract is essential for product quality and effectiveness.
Testing Methodology
Hypericin was quantified by HPLC (High-Performance Liquid Chromatography) using a validated reverse-phase method after irradiation with white light to ensure specificity. Hypericin content was expressed as the sum of hypericin and pseudohypericin.
HPTLC (High-Performance Thin Layer Chromatography) was performed to confirm the product identity and detect common synthetic dyes, including Brilliant Blue (E133), Sunset Yellow (E110), and Tartrazine (E102). These dyes can artificially inflate hypericin readings in less specific testing methods.
Product Testing Results
Out of 22 tested brands, only NOW met the label claim for total hypericin and contained no synthetic dyes.
Nine brands contained synthetic dyes.
Five brands had no detectable hypericin.
None of the gummy brands had any detectable hypericin.
Non-standardized St. John’s Wort whole herb is reported to have as low as 0.03% hypericin, and of the six products containing the non-standardized material, all had less than this amount.
Importance of Analytical Method Selection
Choosing the correct testing method for St. John’s Wort is critical. Non-specific methods, such as simple colorimetric or spectrophotometric assays, can be fooled by synthetic dyes that mimic the deep red color of hypericin. These approaches may artificially inflate potency results, making products appear compliant when they are not. In contrast, HPLC can separate and identify true hypericin compounds from adulterants, providing an accurate and reliable measure of product quality. NOW’s and Alkemist’s testing both relied on chromatographic specificity, ensuring confidence in the findings.
Conclusion
Bottom line is, only NOW® St. John’s Wort met the label claim for total hypericin and showed no evidence of dye adulteration. Our in-house analytical testing, paired with Alkemist Labs’ independent verification, underscores the importance of scientifically valid methods. NOW will continue to purchase and test products sold on major online platforms and publicly share the results to encourage higher quality standards across the supplement industry.
NOW Testing St. John’s Wort Extract Products August 2025
|
Brand of St. John’s Wort Tested |
Size |
Lot # |
Claim per cap/tab/gummy |
Hypericin |
Dyes |
TLC ID |
Score |
|
Blossom Nature |
120 Veg Caps |
Missing |
450mg/0.3% hypericin (1.35 mg) |
0.2 mg |
Detected |
Pass |
Fail |
|
Brieofood |
180 Gel Caps |
12501 |
300mg/0.3% hypericin (0.9 mg) |
0.4 mg |
None detected |
Pass |
Fail |
|
Bronson 700mg (Not standardized) |
180 Veg Caps |
F2502261 |
50mg |
0.1 mg |
None detected |
Pass |
Fail |
|
Bulk Supplements (Not standardized) |
240 Veg Caps |
VC2419407 |
600mg |
0.0 mg |
None detected |
Pass |
Fail |
|
duwhot Gummies |
60 Gummies |
NL25F03 |
300mg/0.3% hypericin (0.9 mg) |
0.0 mg |
Detected |
Fail |
Fail |
|
Health’s Harmony (with 200 mg Herb) |
180 Veg Caps |
224009 |
300mg/0.3% hypericin (0.9 mg) |
0.5 mg |
None detected |
Pass |
Fail |
|
Horbaach 300mg |
120 Gel Caps |
529092 |
300mg/0.3% hypericin (0.9 mg) |
0.3 mg |
Detected |
Pass |
Fail |
|
JOVITHEA Gummies 700mg |
60 Gummies |
Missing |
350mg/0.3% hypericin (1.05 mg) |
0.0 mg |
None detected |
Fail |
Fail |
|
Natures Truth 600mg |
90 Gel Caps |
533517 |
300mg/0.3% hypericin (0.9 mg) |
0.3 mg |
Detected |
Pass |
Fail |
|
Naturebell (Not standardized) |
240 Gel Caps |
0NB093A |
300mg |
0.1 mg |
None detected |
Pass |
Fail |
|
NOW Foods |
100 Veg Caps |
3369738 |
300mg/0.3% hypericin (0.9 mg) |
0.9 mg |
None detected |
Pass |
Pass |
|
NusaPure 6,000mg |
240 Veg Caps |
2407039 |
150mg/0.3% hypericin (0.45 mg) |
0.1 mg |
Detected |
Pass |
Fail |
|
NusaPure 10,000mg |
200 Veg Caps |
2408024 |
500mg/0.3% hypericin (1.5 mg) |
0.2 mg |
Detected |
Pass |
Fail |
|
Nutricost (Whole Herb) |
120 Veg Caps |
25012820 |
500mg |
0.1 mg |
None detected |
Pass |
Fail |
|
O!Vita 300mg Extract (with 200mg Herb) |
100 Tablets |
52493 B |
300mg/0.3% hypericin (0.9 mg) |
0.6 mg |
None detected |
Pass |
Fail |
|
Pure Mountain Botanicals (with 150mg Herb) |
90 Veg Caps |
202418936 |
300mg/0.3% hypericin (0.9 mg) |
0.5 mg |
None detected |
Pass |
Fail |
|
Satoomi (with Whole Herb) |
90 Veg Caps |
MI/SSTJOC21/1-200 |
300mg |
0.0 mg |
None detected |
Fail |
Fail |
|
Sundance Vitamins 300mg |
60 Gel Caps |
530534 |
300mg/0.3% hypericin (0.9 mg) |
0.3 mg |
Detected |
Pass |
Fail |
|
TenMid (with Whole Herb) |
120 Veg Caps |
MI/TSAG12C04/1-300 |
300mg |
0.0 mg |
None detected |
Fail |
Fail |
|
Tinies Bers Gummies |
60 Gummies |
Missing |
250mg/0.3% hypericin (0.75 mg) |
0.0 mg |
None detected |
Fail |
Fail |
|
Toniiq 7000mg 7:1 Extract |
120 Veg Caps |
2501Ca022 |
500mg/0.3% hypericin (1.5 mg) |
0.6 mg |
Detected |
Pass |
Fail |
|
Vitanica (with 100mg Herb) |
90 Veg Caps |
75106 |
300mg/0.3% hypericin (0.9 mg) |
0.5 mg |
Detected |
Pass |
Fail |
|
Zazzee Naturals 8000mg |
150 Veg Caps |
2025-26215 |
400mg/0.3% hypericin (1.2 mg) |
0.6 mg |
None detected |
Pass |
Fail |
Source: https://www.nowfoods.com/healthy-living/articles/now-tests-st-johns-wort-supplements-sold-amazon
NOW’s Testing of Methyl B-12 Brands on Amazon (March 2025)
NOW Foods Testing Program for Mislabeled Supplement Products
NOW continued its award-winning quality testing program with a survey of 25 different brand samples of Methyl B-12 purchased on Amazon in January 2025. Methyl B-12 was chosen for evaluation due to its high cost and the potential failure of its potency label claim. The results once again identified brands not meeting label claims, but fewer than other expensive supplements NOW has tested in the past.
This round of testing also highlighted that because of the inherent complexity of testing Vitamin B-12 across different matrices and dosage forms and the analyte’s instability, even reputable third-party labs may not be able to produce consistent test results. NOW recommends that this material therefore become a priority for collaboration by testing proficiency expert groups.
NOW purchased two bottles of each brand of Methyl B-12 supplement (plus two NOW products) to perform internal testing in NOW’s labs and cross-reference the results with a reputable external laboratory. However, we encountered significant inconsistencies from the external lab indicating a lack of clarity in test methods so chose not to publish those results. The following data reflect NOW’s internal analysis.
Understanding Methylcobalamin
Methylcobalamin is a highly bioavailable form of Vitamin B-12. Other common forms include Cyanocobalamin and Adenosylcobalamin (Dibencozide). Since Vitamin B-12 is nearly absent in plant-based foods, it is often supplemented by vegetarians. Methylcobalamin is preferred for solid dose supplements due to its stability and immediate usability by the body, while Cyanocobalamin is more stable in liquid forms but requires conversion to an active form within the body.
Testing Methodology
All samples were analyzed using High-Performance Liquid Chromatography (HPLC) at NOW’s laboratories, following a validated method aligned with the USP monograph for Methylcobalamin. However, due to the inherent instability of Vitamin B-12 and the difficulty contract labs face in maintaining stable standards and accurate analyte recovery, results may vary across different testing environments. Below are our key observations:
NOW Products Performance:
Both NOW products tested well above the label claim at 140% and 114%. This is due to intentional overages to ensure full potency at best by date.
Market Performance:
Among the remaining 23 brands, results were highly variable:
11 brands exceeded their label claim, with six testing over 120%.
12 brands tested below 100%, with three showing potency between 90-100%, while three brands contained only 0-1% potency.
Repeat Low Performers:
Monohar, a brand from India, consistently failed, showing 0% potency in this round and during our SAM-e testing in November 2024.
aSquared showed some improvement but only reached 56% potency in its liquid product.
Challenges with Dosage Forms:
Liquid and gummy forms were less stable and harder to analyze accurately compared to tablets and capsules. Only one product met potency regulations, while others ranged from 1% to 105%.
Conclusion
Testing Vitamin B-12 is inherently challenging due to the instability of Methylcobalamin and the difficulty in obtaining consistent results across various matrices and dosage forms. Contract laboratories often struggle with maintaining standard stability and accurate analyte recovery, contributing to the variability observed in this study. Our internal data highlight the industry’s need for more robust testing methods and quality control measures.
NOW Testing Methyl B-12 5,000mcg Products March 2025
⚠︎ = Brands that failed potency testing
|
Brand Tested for Methyl B-12 |
Size |
Lot # |
Label Claim |
NOW Test |
% Results |
|
aSquared Nutrition, FL |
120 Lozenges |
49907 |
5000mcg |
4625mcg |
93% |
|
⚠︎aSquared Nutrition, FL |
2 oz Liquid |
16005 |
5000mcg |
2793mcg |
56% |
|
BestVite, CA |
240 Veg Caps |
30824 |
5000mcg |
6339mcg |
127% |
|
Bronson Vitamins, NY |
60 Lozenges |
F2312067 |
5000mcg |
6654mcg |
133% |
|
⚠︎BulkSupplements.com, NV |
180 Softgels |
SG2409506 |
5000mcg |
4450mcg |
89% |
|
⚠︎CFILIHTC/BHRC Nutrition, NY |
60 Gummies |
JLH24072401 |
2500mcg |
15mcg |
1% |
|
Deal Supplement, CA |
240 Tablets |
Illegible |
2500mcg |
2548mcg |
102% |
|
Doctor’s Recipes, AZ |
90 Tablets |
2406-006 |
5000mcg |
6184mcg |
124% |
|
Horbaach, NV |
120 Lozenges |
527071 |
5000mcg |
6965mcg |
139% |
|
Innate Vitality, CA |
90 Tabs |
2408-007 |
5000mcg |
6123mcg |
122% |
|
Lovita Food, CA |
120 Lozenges |
AC2042 |
5000mcg |
4536mcg |
91% |
|
MicroIngredients, CA |
240 Lozenges |
B10V24 |
2500mcg |
2792mcg |
112% |
|
⚠︎Monoherb, India |
90 Veg Caps |
MH/VB12MEC5/0924 |
5000mcg |
0mcg |
0% |
|
⚠︎Nextdia, CA |
90 Tablets |
B12TS8 |
10000mcg |
0mcg |
0% |
|
NOW Foods |
60 Lozenges |
3359018 |
5000mcg |
6980mcg |
140% |
|
NOW Foods |
90 Veg Caps |
3368471 |
5000mcg |
5689mcg |
114% |
|
NusaPure/Gmax, NC |
150 Veg Caps |
2409037 |
5000mcg |
5205mcg |
104% |
|
Nutricost, UT |
240 Veg Caps |
24091039 |
5000mcg |
5335mcg |
107% |
|
⚠︎Orzax, IL (Turkey) |
120 Tablets |
82320042 |
5000mcg |
4239mcg |
85% |
|
⚠︎Raw Science Liquid, WY |
2 oz Liquid |
5528 |
5000mcg |
2436mcg |
49% |
|
⚠︎Safrel Pharmaceuticals, NJ |
200 Tablets |
A2406 |
5000mcg |
4308mcg |
86% |
|
Seeking Health, WA (2 forms) |
60 Lozenges |
4161200 |
5000mcg |
4775, 1260 |
121% |
|
Vitacook, MN |
90 Tablets |
2407-009 |
5000mcg |
5651mcg |
113% |
|
Vitamatic, NJ |
120 Gummies |
VG240702 |
2500mcg |
2623mcg |
105% |
|
Why Not Natural, TX |
2 oz Liquid |
8450 |
5000mcg |
4794mcg |
96% |
This is the 20th round of market testing that NOW has conducted, with results shared with Amazon and the FDA to address low-potency products and help consumers make informed decisions.
Source: https://www.nowfoods.com/healthy-living/articles/nows-testing-methyl-b-12-brands-amazon
NOW’s Latest Testing of SAM-e Brands on Amazon (November 2024)
NOW’s award-winning quality testing program’s most recent examination, a follow-up survey of SAM-e samples purchased on Amazon in October 2024, demonstrates that profound quality and labeling failings continue.
This is the 19th round of market testing that NOW has performed. As always, the company openly shares these results with Amazon and the FDA hoping they will take action to stop these practices, and with consumers so they can make informed purchases.
NOW previously conducted a survey of suspicious SAM-e (S-adenosylmethionine) supplements purchased on Amazon in March 2020 and found all 11 products tested to be low potency, and two of those brands had zero potency. That was distressing, but not shocking. A lawsuit was filed in 2019 against several similar brands of SAM-e due to low potency, but that was eventually dismissed. (See www.truthinadvertising.org and search for SAM-e).
NOW chose to investigate SAM-e for a second time because the product is expensive, and brands appear to cheat regularly with costly products such as SAM-e. In this reexamination of the SAM-e category, NOW decided to gather a new round of samples from more brands and see if any brands corrected their false labeling claims. In October 2024, NOW purchased two bottles each of 24 suspicious SAM-e supplements on Amazon to test internally and externally at Eurofins labs. Some brands were chosen because they appeared to be mislabeled based on excessively high potency claims. NOW chose several in the gummy format, because SAM-e is unstable and can degrade quickly due to moisture and heat.
Because SAM-e is an unstable compound it is often enteric-coated to maintain stability. Ingredient supplier Gnosis by Lesaffre also makes a proprietary SAM-e from disulfate tosylate salt that is stable enough to avoid enteric coating. SAM-e potency is somewhat confusing to understand. It takes a minimum of 800 mg of S-Adenosyl L-Methionine Tosylate Disulfate to produce 400 mg SAM-e (S-Adenosyl L-Methionine). Some brands label full disclosure to include this, while others do not. It is not required but does help consumers fully understand what is in the product.
The chart below shows five repeat low-potency brands that continue to be widely sold on Amazon. Note that 48% of failing samples were found in Florida-based brands with some sharing matching or near-identical lot numbers. All samples were tested by High-Performance Liquid Chromatography (HPLC) at both NOW’s and Eurofins’ labs using a validated method for testing SAM-e based on the USP monograph for SAM-e.
Here are some conclusions:
NOW’s product tested well above the label claim at 109% average. NOW adds significant overages to ensure full potency at expiration date.
20 of the remaining 23 samples failed potency testing (highlighted red in the table), and 16 of the remaining 23 brands contained less than 20% of labeled potency! Six brands contained ZERO potency at all, including both Gummy products.
The five brands that failed testing in 2020 (highlighted yellow in the table) failed again, with one containing zero potency and three containing less than 20% of label claim. Repeated failures include aSquared, Healthy Way, Mono Herbs, Nasa B’Ahava, and Superior Health. Mono Herbs tested at zero potency for the second time in four years.
Ojos Labs and Shevat Vitamins are located in Florida, and their samples share the same lot number. Both, likely made by the same manufacturer, contained less than 10% potency.
aSquared brand is a repeat potency failure. In the past six years, NOW has tested 13 different lots of different products by aSquared, and every single product tested failed potency testing. This product was below 20% potency.
The Natures Craft and Phytoral brands also had nearly identical lot numbers. Both tested below label claims on average but were close to full potency.
The Spliferkou brand makes ridiculous potency claims. This product title claims to include an impossible 3,000 mg, and that is before doubling the SAM-e potency input needed to reach 1800 mg claim. Typically, large double 00 capsules can include a maximum of 1,000 mg, while adding up the claimed ingredients alone nets 2,350 mg per capsule. There is no physical way to reach the levels claimed for this product. Not surprisingly, this brand tested with zero SAM-e potency.
One brand labeled in vegetarian capsules tested to contain animal gelatin. This was Florida Herbal Pharmacy, which also failed with zero potency.
NOW Testing SAM-e Products November 2024
⚠︎ = Brands that failed potency testing
⚠︎⚠︎ = Brands that failed potency testing in 2020 as well as 2024
SAM-e Amazon Products Tested November 2024
|
Brand of SAM-e |
Size |
Lot # |
Potency Claim |
NOW Results |
Eurofins Results |
Avg. Results |
|
⚠︎ Actif American Health |
60 Caps |
Illegible |
400 mg |
46 |
29 |
37 |
|
⚠︎⚠︎ aSquared, FL |
90 Caps |
15672 |
400 mg |
72 |
68 |
70 |
|
Bestvite, CA |
60 Caps |
60524 |
200 mg |
225 |
203 |
214 |
|
⚠︎ Bulk Supplements, NV |
120 Caps |
GC2403502 |
400 mg |
181 |
172 |
177 |
|
⚠︎ Clear Formulas, FL |
90 Caps |
150823 |
500 mg |
35 |
27 |
31 |
|
⚠︎ Colorful Energy, NY |
60 Caps |
231216221 |
400 mg |
13 |
11 |
12 |
|
⚠︎ Florida Herbal Pharmacy |
120 Caps |
MI/SAMC/0524 |
500 mg |
0 |
0 |
0 |
|
⚠︎ Healing Awakening, FL |
90 Caps |
140124 |
500 mg |
35 |
31 |
33 |
|
⚠︎⚠︎ Healthy Way, FL |
90 Caps |
100824 |
400 mg |
40 |
37 |
38 |
|
⚠︎ InFusions, FL |
90 Caps |
200824 |
400 mg |
41 |
40 |
40 |
|
⚠︎ Jovianoy Gummies |
90 Each |
60151DGJ-946 |
533 mg |
0 |
0 |
0 |
|
⚠︎ Kanacoufce, CA |
60 Tabs |
HRW-01897391 |
600 mg |
0 |
0 |
0 |
|
⚠︎⚠︎ Mono Herbs UK/India |
90 Caps |
MH/SAMC300/0724 |
300 mg |
0 |
0 |
0 |
|
⚠︎⚠︎ NasaBe’Ahava, FL |
90 Caps |
150324 |
500 mg |
54 |
49 |
51 |
|
⚠︎ Natures Craft, FL |
60 Caps |
0124128 |
200 mg |
183 |
150 |
166 |
|
Natures Trove, NJ |
30 Tabs |
301086 |
400 mg |
415 |
401 |
408 |
|
⚠︎ Newt Nutrition, FL |
90 Caps |
50431 |
500 mg |
105 |
99 |
102 |
|
NOW |
60 Tabs |
3341247 |
400 mg |
450 |
424 |
437 |
|
⚠︎ Ojos Labs, FL |
60 Caps |
040324 |
500 mg |
48 |
46 |
47 |
|
Phytoral, FL |
90 Caps |
0124129 |
200 mg |
203 |
181 |
192 |
|
⚠︎ Shevat Vitamins, FL |
60 Caps |
040324 |
500 mg |
23 |
18 |
20 |
|
⚠︎ SHIZAM Gummies |
60 Each |
BWF6P |
500 mg |
0 |
0 |
0 |
|
⚠︎ Spliferkou, CA |
60 Caps |
WY-300-0815 |
900 mg |
0 |
1 |
0 |
|
⚠︎⚠︎ Superior Health, OH |
90 Caps |
2202041 |
200 mg |
118 |
66 |
92 |
NOW does this testing to publicly report which brands are labeling accurately. We welcome brands to communicate with NOW about these findings and openly share this information with all customers, industry trade groups and FDA.
Source: https://www.nowfoods.com/healthy-living/articles/nows-latest-testing-sam-e-brands-amazon
NOW Tests Resveratrol Products Purchased Online, Reveals Significant Problems (August 2024)
NOW has continued the testing program on products purchased online, with the company’s state of the art labs recently investigating 30 Resveratrol supplements purchased on Amazon. This is NOW’s 18th round of product testing since 2017, all of which have raised serious concerns.
“Buyer beware” certainly applies to resveratrol, as we found significant problems with most products tested,” said Dan Richard, NOW Health Group VP of Global Sales and Marketing. “Once again, NOW will share this info with Amazon directly and with the FDA, in hopes of further action.”
This popular antioxidant supplement was first isolated in 1939 and has been sold by NOW since 2005. It is a polyphenol naturally found in grapes, red wine, peanuts, and herbs such as Japanese knotweed roots. There are two types of resveratrol: cis-resveratrol and trans-resveratrol. Both have the same molecular formula, but trans-resveratrol is more bioavailable and easier for the body to absorb. Japanese knotweed extract is the most common form of resveratrol in dietary supplements, and the trans-resveratrol portion is often found between 50-98%. Natural resveratrol also contains many trace levels of additional antioxidants, while synthesized resveratrol is isolated chemically and does not contain other synergistic components.
NOW bought two bottles of each product from 30 brands, including NOW, on Amazon. Even before testing, most products seemed suspicious due to inflated potencies and mislabeling on the front label panels.
In addition to in-house testing, NOW sent one unopened bottle of each brand to Alkemist Labs, an industry-leading contract laboratory NOW has partnered with previously, for an independent report on the same products. Both NOW and Alkemist performed high-performance liquid chromatography (HPLC) testing. High-performance liquid chromatography (HPLC) with ultraviolet light detection (HPLC-UV) is the correct method to accurately measure the amount of trans-resveratrol in a sample. Resveratrol is a very interesting natural compound to test due to its complexity and labeling options. It’s important to note that labeling for resveratrol dietary supplements is inconsistent across different brands.
Below are various examples of Supplement Facts panels to illustrate the extent of these differences and the potential confusion they may cause. NOW found two-thirds of the 30 brands tested have confusing or dishonest labeling for Resveratrol. They also found that 21 out of 30 brands tested were below labeled potency, and 17 contained less than 10% of claimed potency. Many brands display the highest potency number on the front panel and in Amazon search terms, but “tricky” labeling on the side panel may not match numbers.
- Both aSquared Nutrition Resveratrol Ultra Strength and NasaBe’Ahava Resveratrol 1000 mg claim to contain 1,000 mg of Resveratrol on the front panel. However, they only provide “8% potency yield” per 2 capsules, resulting in just 40 mg of Resveratrol per capsule, significantly lower than the claimed amount. Although aSquared Nutrition’s test results indicated a higher amount than 40 mg, the initial claim remains deceptive. It is worth noting that aSquared Nutrition has failed NOW’s testing program 12 times out of 12 different products, resulting in a 100% failure rate.
- Purify Life Resveratrol Gummies claims “1500 mg per serving” on the front panel, but only 10 mg of 150:1 extract in the Supplements Facts panel. This is deceptive and improper labeling.
- The front panel of the July 69 brand indicates that it contains 600 mg of Resveratrol per serving. However, the side panel lists the ingredients as “Resveratrol Blend 600 mg” and includes additional complicated information such as “Japanese Knotweed (10% Trans-Resveratrol) (Polygonum cuspidatum)(root), Japanese Knotweed (50% Trans-Resveratrol)(Polygonum cuspidatum)(root)” in very small font size. This labeling might be confusing for some people who are trying to understand what these percentages and ingredients mean.
- The side panel of Dr. Martin’s Nutrition Resveratrol lists “Trans-Resveratrol Complex” as 920 mg per two capsules, combined with nine other fruits and herbs. The side panel has an extremely small font size and is barely legible. The high claimed potency, unknown brand, and misleading labeling led us to include this product in testing.
NOW HPLC Testing Resveratrol Supplements August 2024
|
Brand Tested for Resveratrol |
Size |
Lot # |
Label Claim/Cap |
Honest Labeling |
NOW Test (mg/cap) |
Alkemist Test (mg/cap) |
Avg % of Label Claim |
|
aSquared FL |
180 Veg Caps |
RE9997-320-2A |
500 mg/8% |
No |
43 |
44 |
9% |
|
biodora Formula, GA |
180 Veg Caps |
BR675131-L2 |
400 mg Trans |
No |
19 |
17 |
5% |
|
Clean Nutra Formula, CA |
60 Capsules |
VM2300316/1 |
300 mg |
No |
2 |
1 |
0% |
|
Doublewood Supplements, PA |
120 Veg Caps |
2321501 |
250 mg/125 mg |
Yes |
122 |
130 |
100% |
|
Dr. Martins Nutrition, NV |
180 Veg Caps |
R23354002 |
460 mg Trans |
No |
35 |
36 |
8% |
|
EN Health, NY |
90 Gummies |
384248010901 |
750 mg/98% |
No |
6 |
5 |
1% |
|
Fito Medic’s, Columbia |
180 Capsules |
FEXPO194 |
45,000 mg |
No |
133 |
137 |
0% |
|
GeneX Formulas, FL |
90 Veg Caps |
RE8057-170-1 |
500 mg/50% |
No |
242 |
256 |
50% |
|
GriMed Formula, KY |
90 Veg Caps |
230304 |
200 mg Trans |
No |
10 |
11 |
5% |
|
July 69, WY |
60 Veg Caps |
RVT231110-60 |
600 mg Blend |
No |
152 |
156 |
Inconclusive |
|
MAAC10 FL |
60 Veg Caps |
M232684S |
250 mg Trans |
Inconclusive |
238 |
246 |
97% |
|
MECISCO Formula, OR |
90 Veg Caps |
M/MRES89C04/1-1000 |
500 mg Trans |
Inconclusive |
3 |
0 |
0% |
|
MyPrimaVita Formula, GA |
90 Veg Caps |
PR6624-071-L1 |
333 mg Trans |
No |
14 |
16 |
5% |
|
NasaBe’Ahava, FL |
180 Veg Caps |
130923 |
1000 mg/8% |
No |
45 |
43 |
4% |
|
Nature Bell, CA |
180 Veg Caps |
P2311016 |
333 mg Trans |
Yes |
288 |
303 |
89% |
|
NOW Foods, IL |
60 Veg Caps |
3339738 |
700 mg/350 mg |
Yes |
386 |
412 |
114% |
|
Nutricost, Utah |
120 Veg Caps |
24031315 |
700 mg/350 mg |
Yes |
365 |
359 |
103% |
|
NutriVein Formula, WA |
120 Veg Caps |
NV1023 |
302 mg |
No |
30 |
32 |
10% |
|
Purely Beneficial Formula, CA |
180 Veg Caps |
NR6844-143-L1 |
302 mg |
No |
5 |
6 |
2% |
|
Purest Vantage, FL |
60 Veg Caps |
RE6185-029-L1 |
300 mg/50% |
No |
157 |
144 |
50% |
|
Purify Life Gummies, IL |
60 Gummies |
215011001R |
5 mg |
No |
5 |
6 |
100% |
|
Purity Labs, CA |
90 Veg Caps |
A24B011 |
675 mg Trans |
No |
10 |
13 |
2% |
|
Simple Organics, DE |
90 Veg Caps |
SR6790-117-L1 |
400 mg Trans |
No |
14 |
16 |
4% |
|
Super Natural GOODS, FL |
60 Veg Caps |
Illegible |
302 mg Trans |
Inconclusive |
334 |
341 |
112% |
|
Sylan, TX |
180 Veg Caps |
RAR1008 |
500 mg |
No |
44 |
51 |
10% |
|
Teaveli12 hour SR, 2.5X, CA |
30 Veg Caps |
142301023 |
333 mg |
Inconclusive |
107 |
101 |
31% |
|
Thomas all-natural Remedies |
120 Veg Caps |
TR10427 |
400 mg Trans |
No |
27 |
28 |
7% |
|
Toniiq, IL |
60 Veg Caps |
23L0051 |
300 mg Trans |
Yes |
307 |
322 |
105% |
|
We Like Vitamins, TX |
180 Veg Caps |
60231 |
500 mg/8% |
No |
40 |
43 |
8% |
|
Zetelixia Gummies, CA |
60 Gummies |
Illegible |
750 mg Trans |
Inconclusive |
0 |
0 |
0% |
(Note: Regarding the discrepancy in lab-to-lab results, there will always be a level of deviation between labs due to various factors such as method differences, uncertainty, different analysts, instrumentation, etc. Even within the same lab, results are rarely identical.)
Both labs also used high-performance thin-layer chromatography (HPTLC) for internal testing to check if Japanese knotweed was used as a source of resveratrol. They compared the samples with verified Japanese knotweed reference material and assessed their similarity. This testing was carried out at Alkemist Labs alongside the HPLC testing. Most of the samples contained resveratrol extract with a purity of over 98%. In these cases, neither could confirm the botanical source. The results from both labs are shared below:
NOW HPTLC Testing Resveratrol Supplements August 2024
|
Brand Tested for HPTLC |
NOW HPTLC Result |
Alkemist HPTLC Result |
|
aSquared FL |
Passes |
Passes |
|
biodora Formula, GA |
Fails |
Fails |
|
Clean Nutra Formula, CA |
Fails |
Fails |
|
Doublewood Supplements, PA |
Passes |
Passes |
|
Dr. Martins Nutrition, NV |
Fails |
Fails |
|
EN Health, NY |
Cannot verify botanical source |
Cannot verify botanical source |
|
Fito Medic’s, Columbia |
Cannot verify botanical source |
Cannot verify botanical source |
|
GeneX Formulas, FL |
Passes |
Passes |
|
GriMed Formula, KY |
Cannot verify botanical source |
Cannot verify botanical source |
|
July 69, WY |
Fails |
Fails |
|
MAAC10 FL |
Cannot verify botanical source |
Fails |
|
MECISCO Formula, OR |
Fails |
Fails |
|
MyPrimaVita Formula, GA |
Passes |
Passes |
|
NasaBe’Ahava, FL |
Passes |
Passes |
|
Nature Bell, CA |
Cannot verify botanical source |
Cannot verify botanical source |
|
NOW Foods, IL |
Passes |
Passes |
|
Nutricost, Utah |
Passes |
Passes |
|
NutriVein Formula, WA |
Fails |
Fails |
|
Purely Beneficial Formula, CA |
Fails |
Fails |
|
Purest Vantage, FL |
Passes |
Passes |
|
Purify Life Gummies, IL |
Cannot verify the botanical source |
Cannot verify the botanical source |
|
Purity Labs, CA |
Passes |
Passes |
|
Simple Organics, DE |
Passes |
Passes |
|
Super Natural GOODS, FL |
Cannot verify the botanical source |
Cannot verify the botanical source |
|
Sylan, TX |
Passes |
Passes |
|
Teaveli12 hour SR, 2.5X, CA |
Cannot verify the botanical source |
Cannot verify the botanical source |
|
Thomas all-natural Remedies |
Passes |
Passes |
|
Toniiq, IL |
Cannot verify the botanical source |
Cannot verify the botanical source |
|
We Like Vitamins, TX |
Passes |
Passes |
|
Zetelixia Gummies, CA |
Fails |
Fails |
“The results above further substantiate the fact that the market is saturated with unscrupulous companies that make false claims,” Richard said. “Consumers should be cautious when buying from lesser-known brands and opt for trusted, honest, and transparent brands.
46% of Creatine Gummies Fail Label Claim (February 29, 2024)
In its latest round of testing competing supplement, supplement company NOW found widespread failings to meet label claims when testing several creatine gummies it purchased online. This exercise reached a second red flag when, following the testing program’s usual practice of simultaneous testing by a reputable third-party lab, none of the outside labs NOW vetted and approved were capable of testing gummies.
A survey of several brands of creatine was performed to understand the quality available on the marketplace, which included the brands Astro Labs, Beast Bites, Create, Con-Cret, Greabby, and Njord who failed to meet the label claim, resulting in a 46% failure rate.
Gummy brands testing below label claim were also tested for creatinine using HPLC. Several creatine gummies were found to contain significant amounts of this unwanted creatine metabolite, while also not meeting their claimed creatine strength. Creatinine is a waste product that naturally builds up in blood when muscles are exercised. Bodies produce creatinine at a constant rate, and kidneys usually eliminate almost all of it. Having very high or low creatinine levels can be a health concern; creatinine supplements are not recommended. Creatine in powder form is stable, but when mixed with water can turn into creatinine. Gummies are not an ideal form for creatine supplements because water is used to make gummies, so it can be difficult to get the correct dosage of creatine, NOW reports.
Astro Labs, Greabby, and Njord had a small amount of creatinine detected, while Beast Bites, Create, and Con-Cret had large amounts of creatinine present. Taking the creatine and creatinine data together shows that Astro Labs, Greabby, and Njord likely had a minimal amount of creatine, almost all of which converted to creatinine.
Source: https://www.nutraceuticalsworld.com/contents/view_breaking-news/2024-02-29/now-reports-widespread-failings-in-creatine-gummy-tests/?
26 of 33 Berberine Products on Amazon and Walmart.com Don’t Contain Labeled Amount of Berberine (December 20, 2023)
In the 16th round of product testing since 2017, NOW tested more than 30 Berberine supplements after surveying the online marketplace for questionable products and identified serious quality problems from “no name” brands sold on Amazon and Walmart.com.
- The products chosen were purchased from both Amazon and Walmart.com in early November 2023. These brands were picked because they are less known and sold almost entirely on these platforms. (We have chosen not to test health food store brands or practitioner brands as our experience shows them to be less of a concern.)
- The results of this round of testing showed serious levels of low potencies with every brand testing below 100% potency, except NOW. Seven brands did contain over 80% labelled potency and three brands contained 90-97% potency.
- NOW has tested some of the same brands previously, while testing other categories, and found similar problems.
- Eighteen of the 33 brands tested contained less than 40% of labelled potency. That’s more than half of all brands tested that didn’t even contain a mediocre 40% level of potency.
- Seven of the 33 tested samples had 1% or less of Berberine potency in each product. All of these seriously flawed brands were tested for the first-time in NOW’s testing programme and may be new brands.
Source: https://nutraceuticalbusinessreview.com/berberine-test-results-reveal-serious-quality-problems-from
14 of 21 Astaxanthin Products on Amazon Contain Lower than Labeled Amount (August 24, 2023)
We performed a preliminary investigation into the identity of the 14 brands failing NOW Foods astaxanthin potency testing… so here’s a quick and dirty breakdown of the cheaters.
1. About half of the brands are not associated with a physical address.
And out of the ones I could find a physical address for, only one address has a building large enough to produce supplements. According to Google Maps, most brands that had a physical address were located inside an unrelated business, or a residence like an apartment or house.
2. About half of the brands have no website.
Those without websites only have an Amazon Seller and/or Walmart seller page with no contact information.
3. Thirteen of fourteen brands don’t list the name of an owner or person responsible.
(Don’t you hate when the “About Us” page says nothing about who’s behind the brand, and the “Contact Us” page has no address or phone number? Too bad, because that’s the new normal.)
4. ZERO Amazon-only Seller pages listed a person’s name, business address or phone number.
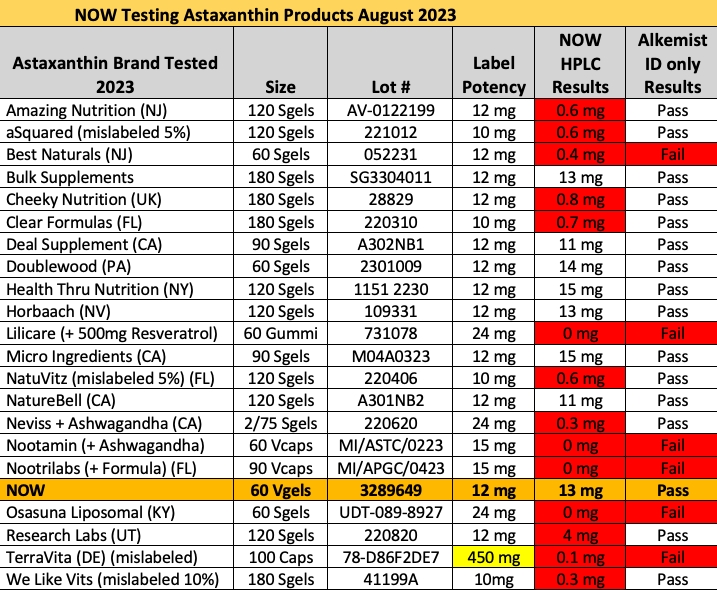
As reported in Nutraceuticals World in August 2023, 13 of 22 supplement brands tested by NOW Foods from Amazon Sellers contain less than 10% of the labeled amount of astaxanthin.
Astaxanthin is generally an expensive ingredient, and manufacturers are often interested to cut costs on testing or production when no one is looking.
AMAZON ASTAXANTHIN PRODUCT MISLABELED RESULTS, PER SERVING
Amazing Nutrition: contains 0.6 mg versus 12 mg labeled
aSquared: contains 0.6 mg versus 10 mg labeled
Best Naturals: contains 0.4 mg versus 12 mg labeled
Cheeky Nutrition: contains 0.8 mg versus 12 mg labeled
Clear Formulas: contains 0.7 mg versus 12 mg labeled
Lilicare: contains 0 mg versus 24 mg labeled
NatuVitz: contains 0.6 mg versus 10 mg labeled
Neviss: contains 0.3 mg versus 24 mg labeled
Nootamin: contains 0 mg versus 15 mg labeled
Nootrilabs: contains 0 mg versus 15 mg labeled
Osasuna: contains 0 mg versus 24 mg labeled
Research Labs: contains 4 mg versus 12 mg labeled
Terra Vita: contains 0.1 mg versus [incorrect dosage] labeled
We Like Vitamins: contains 0.3 mg versus 10 mg labeled
15 of 20 Bromelain Supplements Fail Potency (March 1, 2023)
- 15 out of 20 samples tested (75%) failed to meet label claims for potency. Only six out of 20 samples contained over 50% of labeled potency.
- Shocking that 12 out of 20 brands contained less than even 10% of label claim! That means more than half of all tested Bromelain supplement samples contained less than one-tenth of label claims — the most egregious testing results NOW has seen!
- Three samples had ambiguous labeling that only claimed weight and no activity. Generally, Bromelain powder has an activity of 2,400 GDU per gram, but these brands made no potency claims. Two of these products’ potencies were below the detection limit and were reported as < 10 GDU/g. The third brand, Cadane, only contained 34 GDU of Bromelain per capsule, which is extremely low potency.
- Three brands were labeled as products made in India. These brands tested to only contain 10, 34 and 78 GDU potencies, respectively. Two were under 10% of label claim and the third made no potency claim.
- Only four brands exceeded 100% potency with NOW as one at 121%.
- NOW intentionally did not test known “natural” or practitioner brands. The reason being these other brands typically abide by Good Manufacturing Practices (GMPs) and we expect full potencies.
- NOW has been testing products sold by lesser-known brands for six years and consistently finds most suspect products failing potency testing. We have shared this information widely, with the goal of addressing bad quality products in the marketplace. Unfortunately, testing shows the problem is getting worse.
- NOW has been testing products exclusively found on Amazon, but we will begin adding unknown products found on Walmart.com in the future. We are finding that low potency products often spend the most dollars marketing on both Amazon and Walmart.com. Two of the top four “sponsored” or paid advertised brands from a search on Amazon in March 2023 included the violating brands below.
⚠︎ = Brands that had 100%+ potencies
|
Brand Tested for Potency |
Size |
Lot # |
Label Potency |
GDU Claim |
Total GDU/Cap |
Test Results |
% Label Claim |
|
Amazing Nutrition (NJ) |
120 Vcaps |
AV-21120??? |
500mg |
2,400 |
1,200 |
24 GDU/cap |
2% |
|
Balance Greens (MI) |
180 Caps |
VM2200267/1 |
500mg |
2,400 |
1,200 |
12 GDU/cap |
1% |
|
Best Naturals (NJ) |
120 Tabs |
12251 |
500mg |
600 |
300 |
17 GDU/tab |
6% |
|
Best Vite (CA) |
120 Vcaps |
110621 |
500mg |
2,400 |
1,200 |
96 GDU/cap |
8% |
|
Cadane (India) + Formula |
60 Vcaps |
CRBO6C01/1 |
300mg |
? |
? |
34 GDU/cap |
n/a |
|
Deal Supplement (CA) |
180 Caps |
21-388 |
500mg |
300 |
150 |
126 GDU/cap |
84% |
|
Earthborn Elements (OR) |
200 Caps |
76255 |
575mg |
? |
? |
< 10 GDU/cap |
0% |
|
Fresh Nutrition (CA) |
90 Vcaps |
B21224006-1 |
208mg |
2,400 |
500 |
608 GDU/cap |
122% |
|
H&C (India) |
180 Vcaps |
NCBE102 |
250mg |
2,400 |
600 |
10 GDU/cap |
2% |
|
⚠︎ Horbaach |
120 Caps |
123369 |
850mg |
600 |
480 |
690 GDU/cap |
144% |
|
Herbal Secrets (NJ) |
120 Tabs |
AN-22101702 |
500mg |
300 |
150 |
37 GDU/cap |
25% |
|
⚠︎ NOW |
120 Vcaps |
3277931 |
500mg |
2,400 |
1,200 |
1451 GDU/cap |
121% |
|
⚠︎ Nutricost (UT) |
120 Vcaps |
22102614 |
500mg |
2,400 |
1,200 |
1202 GDU/cap |
100% |
|
NutriONN (OR) |
120 Vcaps |
B989710 |
500mg |
2,400 |
1,200 |
39 GDU/cap |
3% |
|
NUSA Pure/GMAX (NC) |
150 Vcaps |
2206038 |
750mg |
300 |
225 |
20 GDU/cap |
9% |
|
Pure Supplements (UT) |
100 Caps |
73628 |
575mg |
? |
? |
< 10 GDU/cap |
0% |
|
⚠︎ Superior Labs (UT) |
120 Vcaps |
2202025 |
500mg |
2,400 |
1,200 |
1321 GDU/cap |
110% |
|
TenMid (India) + Formula |
90 Vcaps |
TBRO9C01/1 |
350mg |
2,400 |
840 |
78 GDU/cap |
9% |
|
Vitamatic (NJ) |
180 Caps |
P2100020 |
500mg |
2,400 |
1,200 |
815 GDU/cap |
68% |
|
We Like Vitamins (TX) |
180 Vcaps |
BR8958-138-3 |
500mg |
2,400 |
1,200 |
< 10 GDU/cap |
0% |
Source: https://www.nowfoods.com/quality-safety/testing-results-bromelain-supplements-purchased-amazon
7 of 8 CoQ10 Supplements Fail Potency Testing (April 2022)
NOW continued its award-winning industry self-policing program of testing unfamiliar brands found on Amazon and, unfortunately, the cheating continues. The program, begun in 2017, tests high value products sold by unheard of brands on Amazon at both internal and external labs, and evaluates the results compared to label claims.
NOW reexamined eight brands of CoQ10 to see if those that were identified as low potency in testing done in 2020 had improved and found the same serious problems remain for seven out of eight brands tested. Additionally, as shown below, NOW found brands cheating by misrepresenting potencies through deceptive labeling tricks.
NOW purchased three samples of each product below and tested by HPLC both internally at NOW’s state-of-the-art labs and externally at the highly regarded Eurofins labs. It is apparent by looking at lot numbers and bottle types that the same manufacturer is supplying multiple brands with the same fraudulent products (see Florida brands in the chart below).
- Clear Formulas, aSquared, Foxy Doc and Healthy Way brands all mislabel their product as “400mg/6%” potency. This is deceptive when the front panel says “400mg” potency and the Amazon title says “CoQ10 400mg Max Strength”. The customer gets less than 24mg CoQ10 per capsule.
- NOW previously tested a variety of CoQ10 brands on Amazon in 2017, 2018 & 2020 with similar failing results. aSquared, Healthy Way, NasaBe’Ahava and We Like Vitamins were all under 35% potency in 2020 as well.
- Seven out of eight brands tested had less than 30% of the potency claimed
- Perhaps most alarming, three of the eight brands claimed to be in vegetarian capsules, but testing both at NOW and at Eurofins confirmed gelatin was used. The failing brands are Clear Formulas, Healthy Way and Sundhed.
Amazon Brands April 2022 Size Lot # Label Claim/Cap NOW Results Eurofins Av %
⚠︎ aSquared Nutrition, FL 100 Vcaps 30465 400 mg/6% 20 mg 23.6 mg Mislabel
⚠︎ Clear Formulas, FL 200 Vcaps 30620 400 mg/6% 23 mg 23.3 mg Mislabel
⚠︎ Foxy Doc, PA 200 Vcaps 30611A 200 mg/6% 9 mg 10.9 mg Mislabel
⚠︎ Healthy Way FL 200 Vcaps 30442 200 mg/6% 11 mg 12.4 mg Mislabel
⚠︎ NasaBe’Ahava, FL 200 Vcaps 30662 200 mg 11 mg 12.1 mg 6%
NutriONN, OR 120 Vcaps CQ210710 200 mg 199 mg 198 mg 99%
⚠︎ SUNDHED, FL 60 Vcaps 30520 400 mg 21 mg 21.8 mg 5%
⚠︎ We Like Vitamins, TX 120 Vcaps CQ60050 200 mg 54 mg 53.4 mg 27%
2022 Results found at NOW Foods website
NOW Foods Testing of Magnesium Supplements from Amazon.com (2022)
Magnesium Glycinate has become a very popular form of Magnesium and we tested 16 total samples. NOW discovered that almost all other brands tested failed to include the chelated magnesium form, as claimed on the label. Magnesium chelates, such as magnesium bisglycinate or glycinate, have excellent water solubility and lack a laxative effect. The fully reacted chelates are better absorbed and more expensive than other forms and thus, are at risk for substitution with lower quality material, such as magnesium oxide and magnesium carbonate, simply blended with glycine.
Various magnesium glycinate products, including two manufactured by NOW, were purchased on Amazon and subjected to testing at our in-house lab, as well as at Eurofins contract laboratory. First, total magnesium content was determined by analyzing the samples using Inductively Coupled Plasma – Optical Emission Spectrometry (ICP-OES). To determine the levels of water-soluble magnesium glycinate, the same ICP-OES technology was used, but instead of acid digestion, a gentle water extraction was applied to all samples. Twelve out of 16 tested products met the label claim when we tested total magnesium content. However, only NOW products met the label claim, when we looked at the soluble (chelated) form of magnesium, suggesting that other brands use non-soluble forms of magnesium in place of the more expensive chelated form. Glycine was also detected in all samples, although the soluble magnesium results clearly suggest that glycine was not (or not entirely) bound with magnesium. Unfortunately, it is known in the industry that many brands either knowingly or unknowingly simply blend glycine with magnesium oxide or carbonate and then label the product as “Magnesium Glycinate”. The difference is that the improperly labeled product is much lower cost and is not a fully reacted or bonded chelate. The results are summarized in the table below.
Additionally, it seems some brands mislabel intentionally in order to get higher label potency claims. Deal Supplement brand claims 750mg of “Magnesium Glycinate” per capsule, while legal labeling should list the elemental dose of Magnesium and not the total weight of a RDI ingredient. Other brands that mislabel in this same way include: Innate Vitality, Naturebell, Terranics and ZYY Nutrition. Most brands label Magnesium Glycinate properly, such as below:
Magnesium (as Bisglycinate)…..125mg or Magnesium (from Glycinate)…..125mg
Or Magnesium 200mg (from 2,000mg Magnesium Bisglycinate) as NOW does for full disclosure of total and elemental mineral weights.
We intentionally did not test most brands that claimed to use Albion Minerals**, which are known to be high quality and specialists in fully reacted Magnesium Glycinate. Magnesium Bisglycinate Chelate powder from Albion Minerals/Balchem contains 10% elemental magnesium. This is why it takes 2,000mg of Magnesium Bisglycinate powder to yield 200mg of elemental Magnesium in NOW brand. Some brands such as Toniiq claim that their Magnesium Glycinate is 20% elemental, but this can only be achieved by using Magnesium and Glycine that are not fully reacted/bonded together. NaturaLife appears to be labeled properly with 18% elemental Magnesium potency with Albion as the source, but this also is a blend – “buffered” – in order to reach the higher potency claim. So NaturaLife is accurate because they include “Magnesium Oxide” in the side panel of ingredients, but also deceptive since the front panel only lists Magnesium Glycinate. Horbaach brand seems to accurately label their side panel as: Magnesium 240mg (from 1,330mg Magnesium Glycinate Chelate – Magnesium Bisglycinate Chelate, Magnesium Oxide). But this is also deceptive since the front panel claims 1,330mg per serving and the side panel states that Magnesium is only 240mg. This form of Magnesium Glycinate contains a blend with Magnesium Oxide and claims to be made “with chelated minerals”, but not 100% chelate.
Test Results December 1, 2022
**https://balchem.com/human-nutrition-health/hnh-products/albion-minerals/
⚠︎ = potency claims not met or poor results
| Brand Tested Mag Glycinate Supplements Dec 2022 | Size | Lot # | /Front Panel Claim | Sice Claim/Cap/Tab | NOW Total Magnesium Results | Eurofins Total Magnesium Results | NOW Soluble Magnesium Results | Eurofins Soluble Magnesium Results |
|---|---|---|---|---|---|---|---|---|
| NOW Mag Bisglycinate Powder | 8 oz | 3256627 | n/a | 250 mg/serv | 273 | 265 | 271 | 260 |
| NOW Mag Glycinate 100mg | 180 Tabs | 3258879 | n/a | 100 mg/tablet | 111 | 110 | 111 | 101 |
| ⚠︎ Deal Supplement CA | 200 Caps | P2207015 | 750 mg | 750 mg/cap | ⚠︎ 131 | ⚠︎ 126 | ⚠︎ 75 | ⚠︎ 63 |
| ⚠︎ Doublewood PA | 180 Vcaps | 220257 | 400 mg | 60 mg/cap | 69 | 71 | ⚠︎ 39 | ⚠︎ 28 |
| ⚠︎ Dr. Martin’s Nutrition NV | 180 Vcaps | 405458 | 425 mg | 212.5 mg/cap | 298 | 285 | ⚠︎ 85 | ⚠︎ 64 |
| ⚠︎ Horbaach NV | 250 Caps | 120520 | 1300 mg | 120 mg/cap | 130 | 128 | ⚠︎ 62 | ⚠︎ 49 |
| ⚠︎ Innate Vitality CA | 120 Vcaps | P220702 | 500 mg | 500 mg/cap | ⚠︎ 65 | ⚠︎ 62 | ⚠︎ 52 | ⚠︎ 38 |
| ⚠︎ Nature’s Branch FL | 200 Tabs | 2220527 | 400 mg | 28 mg/tab | 30 | 30 | ⚠︎ 18 | ⚠︎ 16 |
| ⚠︎ NaturaLife Labs CA | 120 Vcaps | 220808 | 450 mg | 150 mg/cap | 162 | 160 | ⚠︎ 64 | ⚠︎ 58 |
| ⚠︎ Natures Craft NY | 90 Vcaps | 2022-14474 | n/a | 133.3 mg/cap | 172 | 179 | ⚠︎ 14 | ⚠︎ 12 |
| ⚠︎ Naturebell CA | 180 Caps | P2206027 | 500 mg | 500 mg/cap | ⚠︎ 67 | ⚠︎ 73 | ⚠︎ 52 | ⚠︎ 41 |
| ⚠︎ Purely Holistic UT | 270 Tabs | 2022-14260 | 400 mg | 200 mg/tab | 196 | 197 | ⚠︎ 47 | ⚠︎ 44 |
| ⚠︎ Terranics CA | 120 Vcaps | P2201007 | n/a | 500 mg/cap | ⚠︎ 47 | ⚠︎ 50 | ⚠︎ 47 | ⚠︎ 40 |
| ⚠︎ Toniiq IL | 240 Caps | 22F0021 | n/a | 150 mg/cap | 167 | 178 | ⚠︎ 68 | ⚠︎ 49 |
| ⚠︎ Whollium MA | 90 Tabs | 2207-015A | n/a | 150 mg/tab | 152 | 150 | ⚠︎ 127 | ⚠︎ 111 |
| ⚠︎ ZYY Nutrition CA | 180 Caps | P2203002 | 1000 mg | 500 mg/cap | ⚠︎ 59 | ⚠︎ 65 | ⚠︎ 49 | ⚠︎ 39 |
NOW Foods Testing of Turmeric and Curcumin Supplements (2021)
Continuing the work to disclose alarming quality failings and misleading, inaccurate labels among unfamiliar brands of products purchased on Amazon, NOW has turned their attention to the curcumin/turmeric category, finding similarly poor results.
Having previously tested multiple categories of suspect supplements purchased on Amazon that revealed low potency and poor quality control, NOW recently tested turmeric extracts for potency, heavy metals, labeling accuracy, and potential addition of synthetic curcumin.
NOW assayed 23 unknown brand product samples plus two NOW products, all purchased from Amazon in June 2021. Initially it appeared that the unknown brands tested much better than expected. Only one product clearly failed potency testing and four others tested very low, but without any specific label claim. This initial look was not the end of the story.
Virtually all of the products were labeled such as “Turmeric Curcumin 1650mg” on the front panel, yet the side panel would list 1,500mg as Turmeric Root, 300mg Ginger Root, 150mg Turmeric Extract and 15mg BioPerine PER THREE CAPSULES. That equals to 655mg per capsule and less than 10% of the product is Turmeric 95% Standardized Extract. “This can be perceived as deceptive since many customers do not know the difference between Turmeric, Turmeric Extract, Curcumin Extract, and Standardized 95% Extract,” said Dan Richard, NOW’s Vice President of Global Sales and Marketing.
NOW also tested heavy metals (Arsenic, Cadmium, Lead, and Mercury) to compare each product. The average total heavy metals, by product, were 525% higher than NOW’s two sample average, and only one product out of 23 had less heavy metals than NOW. Two products, B’Leaf Nature and Eagle brands, were more than 20 times higher than NOW and above California’s Prop 65 limits for Lead. Two others, Farm Haven and BioEmblem brands, were both above 100ppb in Cadmium, a particularly toxic heavy metal.
Synthetic adulteration was also an issue in these tests. Low quality curcumin (turmeric extract) is known to be spiked with synthetic curcumin in order to meet potency testing. The American Botanical Council highlighted this problem, and quality brands have made sure to avoid it. Synthetic curcumin is derived from petrochemicals, rather than natural Turmeric, at a much lower cost. NOW sent all samples to the University of Georgia’s Center for Applied Isotope Studies for independent radiocarbon testing. This lab found that four out of 23 unfamiliar brands were spiked, with “fossil fuel derived organic carbon.” These four brands are: Vitpro, Me First Living, Eagle, and Primal Harvest.
NOW also suspected that some of these products may be mislabeled as Vegetarian Capsules. Of the 23 samples below, two tested to be in animal gelatin capsules and were not in Vegetarian Capsules. These are Bioganix and Nutriflair brands with below lot numbers.
All of these products were tested for potency both at the company’s highly respected internal labs and at Eurofins Labs. The assay method is RP-HPLC with UV detection and potencies are determined based on total curcuminoids per capsule.
In total, 12 out of 23 outside products tested failed either for potency, containing synthetic curcuminoids, heavy metals or used gelatin caps instead of the claimed veggie caps. Somewhat surprisingly, 11 out of 23 did pass all tests, though with slightly misleading labeling.
NOW is the only brand to list both total and net weights of 95% extract on labels. NOW’s # 4638 is labeled showing 665mg Turmeric Extract providing 630mg or 95% Curcumin as the primary active potency.
“While we appreciate Amazon’s initial efforts to address these ongoing, egregious problems with sellers on their platform, there is clearly still a long way to go,” Richard said. “The kind of results we found are not what consumers expect when they purchase dietary supplements from sellers they trust.”
⚠︎ = potency claims not met or poor results
| Curcumin/Turmeric 95% Tests by NOW July 2021 | Size | Lot # | Expir. Date | Label Claim 95% Extract | NOW Results # 1 Test | Eurofins Results | Av % Result |
|---|---|---|---|---|---|---|---|
| NOW Turmeric Curcumin #4638 | 60 Vcaps | 3187526 | 12/2024 | 665 mg | Pass | Pass | 107% |
| NOW Turmeric Curcumin Softgels #4936 | 60 Gels | 3206552 | 4/2024 | 475 mg | Pass | Pass | 102% |
| Ace Nutrition NJ Turmeric Curcumin | 90 Vcaps | 2020-07476 | 06/2023 | 200 mg | Pass | Pass | 102% |
| BioEmblem/ANZA NY Turmeric Curcumi | 90 Vcaps | 1B2201 | 02/2022 | 50 mg | Pass | Pass | 145% |
| ⚠︎ Bioganix FL Turmeric Curcumin | 120 Vcaps | 62053 | 01/2023 | No claim | 13 mg | 10 mg | |
| Bio Schwartz NY Turmeric Curcumin | 90 Vcaps | T20C163 | 12/2022 | 50 mg | Pass | Pass | 141% |
| B’Leaf FL Turmeric Curcumin | 180 Vcaps | 20L0251 | 10/2022 | 50 mg | Pass | Pass | 140% |
| ⚠︎ Doctor Recommended Turmeric Curcumin | 180 Vcaps | TC5151 | None | 208 mg | Fail | Fail | 3% |
| Eagle Supplements PA Turmeric Complex | 60 Vcaps | 2001007 | 01/2022 | 50 mg | Pass | Pass | 98% |
| Farm Haven CA Turmeric Curcumin | 120 Vcaps | 12988 | 07/2022 | 50 mg | Pass | Pass | 140% |
| ⚠︎ FineVine NJ Turmeric Curcumin | 180 Vcaps | None | 01/2024 | Not clear | 21 mg | 21 mg | |
| Me First Living AZ Turmeric Curcumin | 60 Vcaps | 007068 | 11/2022 | 500 mg | OK | OK | 96% |
| MedChoice CA Turmeric Bioperine Garlic | 120 Vcaps | 21C042 | 04/2023 | 50 mg | Pass | Pass | 117% |
| Nature’s Base TX Turmeric & Ginger | 60 Vcaps | 20L044 | 12/2022 | 50 mg | Pass | Pass | 99% |
| Natures Nutrition UT Turmeric Curcumin | 120 Vcaps | 11747-120 | 09/2023 | 50 mg | Pass | Pass | 150% |
| Natures Wellness FL Turmeric Curcumin | 120 Vcaps | A1B006 | 02/2024 | 50 mg | Pass | Pass | 139% |
| Naturewise CA Turmeric Curcumin | 180 Vcaps | T20H075 | 02/2024 | 167 mg | Pass | Pass | 105% |
| New Age Turmeric Curcumin (no address) | 90 Vcaps | 11904 | 02/2024 | 50 mg | Pass | Pass | 162% |
| ⚠︎ NutriFlair WA Turmeric | 180 Vcaps | TC59034 | 02/2024 | Not clear | 8 mg | 8 mg | |
| Primal Harvest NY Turmeric Complex | 60 Vcaps | 012221107 | 03/2023 | 200 mg | Pass | Pass | 119% |
| ⚠︎ Pure by Nature GA Turmeric Curcumin | 180 Vcaps | T58657 | 12/2023 | Not clear | 18 mg | 16 mg | |
| Vimerson Health IL Turmeric Curcumin | 60 Vcaps | 2021-1119 | 03/2023 | 50 mg | Pass | Pass | 142% |
| Vita Breeze FL Turmeric Curcumin | 180 Vcaps | TC59145 | 03/2024 | Not clear | 91 mg | 97 mg | |
| VitPro NJ Turmeric Curcumin | 90 Vcaps | STTCBLK321 | 03/2023 | 150 mg | Pass | Pass | 113% |
| Viva Naturals NY Organic Turmeric | 90 Tabs | 0540B9 | 09/2021 | 50 mg | Pass | Pass | 137% |
NOW Foods Testing of Phosphatidylserine Purchased on Amazon.com (2021)
Following NOW’s report earlier this year of CoQ10, Acetyl-l-Carnitine and SAMe supplements with alarming quality and labeling problems sold on Amazon, NOW has identified similar serious failings in the Phosphatidyl Serine (PS) category.
NOW purchased and tested a group of PS products on Amazon, choosing this particular supplement for several key reasons. According to SPINS data, NOW is the leading seller of Phosphatidyl Serine in natural food stores, and they know the product well. NOW staff noticed that their PS sales were declining on Amazon, and investigated. They found that the top selling brands on Amazon were offering prices that averaged 70% below NOW’s already discounted prices. Knowing that the prices looked “too-good-to-be-true”, it made sense to test and verify potencies. Because PS is an expensive supplement, the incentive is high for marketing brands to put extra funds into Amazon-sponsored fees. NOW had previously tested examples in this and other categories and found these semi-private label brands sold exclusively on Amazon to display faulty labeling and be of poor quality. When most quality brands sell 100mg PS as the standard potency, it’s questionable to see high potencies such as 400mg or 500mg.
NOW’s initial analysis of simply reading label information found multiple problems. Phosphatidyl Serine is a complicated supplement, which makes it easier to have confusion. Many of the brands included in this report list a high potency, such as 500mg, on the front label (and Amazon’s keyword) panel, yet the side panel often states that amount per 2 or 3 capsules. Others were more deceptive by adding things such as “Phosphatidyl Serine Complex” in order to show a high potency value, even when the actual potency of PS would only be 20% of that number. Here are some examples:
- Absonutrix is deceptively labeled as “Phosphatidylserine Complex 500mg”
- Naturebell is deceptively labeled as “Phosphatidylserine 20% 400mg per 2 Cap serving”. Havasu brand also labels this way “Phosphatidyl Serine 20% 100mg”
- longlifenutri is mislabeled as “Phosphatidylserine 500mg (per 2 caps) (standardized to 20%)”
- NUSA brand label is unclear how much Phosphatidyl Serine is in each capsule
- Both Earth Natural brands mislabel listing Phosphatidyl Serine as 500mg on the front, but only 50mg per Cap on the side fine print
- Correct labeling should be “Phosphatidyl Serine 100mg (from 500mg Sunflower/Soy Lecithin Complex)” if the raw ingredient is 20% PS from Complex, or simply “Phosphatidyl Serine….100mg”
A total of 43 samples were purchased on Amazon, generally three bottles of each brand, and tested to verify results. Three of these were NOW brand and results on these were 100% – 108% of label claims. Of the remaining non-NOW brands, two samples were discarded when results revealed 2.5 times the label potency claim so it was suspected these products were spiked with added Serine in order to fool testing. Two samples passed potency (not included in this report), while 36 samples failed. Of the 36 failures listed below, 17 of these products contained less than 10% of the labeled value.
“That’s a shockingly poor failure rate, but not really a surprise based upon our previous experience testing supplements sold on Amazon,” said Dan Richard, Vice President of Global Sales and Marketing. “Amazon has to find a way to raise the bar on products they sell, especially today with people buying more supplements from Amazon in order to stay healthy.”
The results are reported in the chart below, which NOW tested by HPLC both by their in-house labs and independently at Eurofins Labs (www.eurofinsus.com).
Phosphytidal Serine Assay Results May – July 2020 from Amazon Purchases April 3, 2020 and May 27, 2020.
⚠︎ = potency claims not met or poor results
| Brand Phosphytidal Serine on Amazon | Count | Lot # | Expir. Date | Label Claim per cap | Results per cap | Lab Test | % Label Claim |
|---|---|---|---|---|---|---|---|
| NOW Foods | 60 Vcaps | 3138361 | 12/2021 | 100 mg | 104.4 mg | NOW | 104% |
| NOW Foods | 120 Vcaps | 3146319 | 02/2022 | 100 mg | 100.8 mg | NOW | 100% |
| NOW Foods | 120 Vcaps | 3146319 | 02/2022 | 100 mg | 108 mg | Eurofins | 108% |
| ⚠︎ Absonutrix (mislabeled) | 120 Vcaps | AS101918-1 | 10/2021 | 520 mg | 2 mg | NOW | <2% |
| ⚠︎ Absonutrix (mislabeled) | 120 Vcaps | AS101918-1 | 10/2021 | 520 mg | 5.2 mg | NOW | <2% |
| ⚠︎ Absonutrix (mislabeled) | 120 Vcaps | AS101918-1 | 10/2021 | 520 mg | 5.8 mg | Eurofins | <2% |
| ⚠︎ BoostCeuticals (+ Formula) | 100 Caps | 5121926 | 03/2024 | 167 mg | 12.7 mg | NOW | 8% |
| ⚠︎ BoostCeuticals (+ Formula) | 100 Caps | 27012004 | 03/2024 | 167 mg | 57 mg | Eurofins | 34% |
| ⚠︎ Doublewood Supplements | 120 Caps | 1912697 | 01/2023 | 150 mg | 67.4 mg | NOW | 45% |
| ⚠︎ Doublewood Supplements | 120 Caps | 1912697 | 01/2023 | 150 mg | 48.1 mg | NOW | 32% |
| ⚠︎ Doublewood Supplements | 120 Caps | 1912697 | 01/2023 | 150 mg | 24.3 mg | Eurofins | 16% |
| ⚠︎ Dr. Maxwell (+ Bacopa ext,) | 120 Vcaps | P856510 | 01/2023 | 150 mg | zero | NOW | <2% |
| ⚠︎ Dr. Maxwell (+ Bacopa ext,) | 120 Vcaps | P856510 | 01/2023 | 150 mg | zero | NOW | <2% |
| ⚠︎ Dr. Maxwell (+ Bacopa ext,) | 120 Vcaps | P856510 | 01/2023 | 150 mg | 1.7 mg | Eurofins | <2% |
| ⚠︎ GLS Nutrition Formula/Earth Natural | 200 Caps | 11919 | 08/2021 | 250 mg/50 mg | 31.5 mg | NOW | 13% |
| ⚠︎ Havasu Nutrition + Ginkgo 120 mg | 60 Caps | 1019010 | 10/2021 | 100 mg/20% | 58.8 mg | NOW | 59% |
| ⚠︎ Havasu Nutrition + Ginkgo 120 mg | 60 Caps | 1019010 | 10/2021 | 100 mg/20% | 17.9 mg | Eurofins | 18% |
| ⚠︎ Intelligent Labs | 90 Vcaps | PS55398 | 05/2022 | 100 mg | 17.8 mg | NOW | 18% |
| ⚠︎ Intelligent Labs | 90 Vcaps | PS55398 | 05/2022 | 100 mg | 19.5 mg | NOW | 20% |
| ⚠︎ Intelligent Labs | 90 Vcaps | PS55398 | 05/2022 | 100 mg | 29 mg | Eurofins | 29% |
| ⚠︎ longlifenutri | 180 Vcaps | 6175 | 11/2022 | 250 mg/20% | 68 mg | NOW | 27% |
| ⚠︎ longlifenutri | 180 Vcaps | 6156 | 03/2023 | 250 mg/20% | 31.5 mg | NOW | 13% |
| ⚠︎ longlifenutri | 180 Vcaps | 6156 | 03/2023 | 250 mg/20% | 37.3 mg | Eurofins | 15% |
| ⚠︎ Mental Refreshment (+ Formula) | 200 Caps | 16012005 | 02/2022 | 167 mg | 136.8 mg | NOW | 82% |
| ⚠︎ Mental Refreshment (+ Formula) | 100 Caps | 22012005 | 03/2022 | 167 mg | 47.3 mg | Eurofins | 28% |
| ⚠︎ Mono Herb (plus Flax Powder) | 90 Vcaps | 03/2022 | 200 mg | 13.2 mg | NOW | 7% | |
| ⚠︎ Mono Herb (plus Flax Powder) | 90 Vcaps | 03/2022 | 200 mg | 1.4 mg | Eurofins | <2% | |
| ⚠︎ Naturebell (mislabeled) | 180 Caps | A911NB2 | 11/2021 | 200 mg/20% | 34 mg | NOW | 17% |
| ⚠︎ Naturebell (mislabeled) | 180 Caps | A002MB7 | 02/2022 | 200 mg/20% | 28 mg | NOW | 14% |
| ⚠︎ Naturebell (mislabeled) | 180 Caps | A002MB7 | 02/2022 | 200 mg/20% | 5.6 mg | Eurofins | 3% |
| ⚠︎ Naturetition Formula/Earth Natural | 200 Caps | 416-128 | 04/2022 | 250 mg/50 mg | 11.9 mg | NOW | 5% |
| ⚠︎ Naturetition Formula/Earth Natural | 200 Caps | 416-128 | 04/2022 | 250 mg/50 mg | 3.8 mg | NOW | <2% |
| ⚠︎ Naturetition Formula/Earth Natural | 200 Caps | 416-128 | 04/2022 | 250 mg/50 mg | 2.3 mg | Eurofins | <2% |
| ⚠︎ NUSA (+ Formula) | 200 Vcaps | 2004023 | 04/2023 | 250 mg ? | 31.7 mg | NOW | 13% |
| ⚠︎ NUSA (+ Formula) | 200 Vcaps | 2004023 | 04/2023 | 250 mg ? | 2.4 mg | Eurofins | <2% |
| ⚠︎ Superior Health (+ Formula) | 100 Caps | 12041911 | 08/2023 | 250 mg | 7 mg | NOW | 3% |
| ⚠︎ Superior Health (+ Formula) | 100 Caps | 12041911 | 08/2023 | 250 mg | 15.9 mg | NOW | 6% |
| ⚠︎ Superior Health (+ Formula) | 100 Caps | 12041911 | 08/2023 | 250 mg | 3.9 mg | Eurofins | <2% |
| ⚠︎ We Like Vitamins(+ Formula) | 100 Caps | 11101916 | 11/2023 | 167 mg | 138.3 mg | NOW | 83% |
For more information on the NOW Foods testing program, please visit: https://www.nowfoods.com/healthy-living/articles/now-uncovers-quality-issues-supplement-brands-sold-amazon
Warning Letters, Court Orders and Injunctions
FDA warns consumers not to purchase or use Umary and Amazy products as they may be harmful to your health
[9/5/2024] FDA is warning consumers not to purchase or use Umary and Amazy products, marketed as dietary supplements, as they may be harmful to your health. This is due to potentially dangerous hidden drug ingredients not listed on the product label. FDA urges consumers taking these products to talk to their health care professional if they have any concerns.
These products might also be sold under variations of the name Umary and Amazy. They are promoted to treat pain and other conditions. These products are sold on various websites, including SoloVital, Main Products and Amazon, and in some retail stores.
FDA has received an increase in adverse event reports with the use of Umary products since the agency issued a warning in June 2024. The adverse event reports are consistent with known common adverse reactions such as gastrointestinal, cardiovascular and serious skin reactions associated with the drug ingredients found in Umary.
FDA laboratory testing found certain Umary and Amazy products contain the drug ingredients diclofenac and omeprazole, which are not listed on the product label. Diclofenac is a non-steroidal anti-inflammatory drug (NSAID). NSAIDs may cause increased risk of cardiovascular events, such as heart attack and stroke, as well as serious gastrointestinal damage, including bleeding, ulceration and fatal perforation of the stomach and intestines. Diclofenac also may interact with other medications and significantly increase the risk of adverse events, particularly when consumers use multiple NSAID-containing products.
Omeprazole is a proton pump inhibitor (PPI) used to treat gastric (stomach) acid-related disorders. PPI medicines may cause serious skin reactions, abdominal pain, diarrhea, nausea and headache. Omeprazole may also mask stomach issues such as erosions, ulcers and stomach cancer, and it can also interact with other medications.
The agency recently issued a warning letter to SoloVital for selling Umary. In July 2024, Main Products and SoloVital recalled Umary products.
The agency has seen an increase in fraudulent products marketed for pain that contain hidden ingredients. Consumers should use caution when considering purchasing these types of products as these products may be harmful.
The agency maintains a database of health fraud products and encourages consumers to beware of health fraud scams.
Anthony Trinh, 123Herbals LLC (123HERBALS.COM) Issues Nationwide Recall of Silintan Capsules Due to the Presence of Undeclared Meloxicam
Company Announcement
FOR IMMEDIATE RELEASE – Date 01/07/2026 – Rosemead, CA, ANTHONY TRINH, 123herbals LLC is voluntarily recalling all lots of Silintan capsules to the consumer level. FDA analysis has found the product to be tainted with meloxicam. Meloxicam, is an approved Nonsteroidal Anti-Inflammatory Drug, (NSAID) indicated for management of osteoarthritis pain. Products containing meloxicam cannot be marketed as dietary supplements. The presence of Meloxicam in Silintan capsules, renders it an unapproved new drug for which the safety and efficacy have not been established and, therefore, subject to recall.
Risk Statement: Exposure to meloxicam at doses exceeding the recommended maximum daily dose of 15mg/day, may result in a reasonable probability of serious life-threatening events such as blood clots, heart attack, and stroke. Patients also face risks of hypersensitivity reactions, gastrointestinal bleeding, ulceration, and perforation, as well as acute kidney injury, particularly in elderly. Dangerous drug-drug interactions may occur in patients on blood thinners or other NSAIDs, significantly increasing bleeding risk. To date, Anthony Trinh, 123Herbals LLC has not received any reports of adverse events related to this recall.
The tainted Silintan is marketed as a dietary supplement for relief of joint and body aches and is packaged in 25-count plastic bottles with blue labels (see labels below). The affected Silintan capsules include all lot numbers within expiry. Silintan capsules were distributed nationwide online at 123herbals.com
Anthony Trinh, 123Herbals LLC is notifying its distributors and customers by emails and is arranging for returns of all recalled products. Consumers and retailers that have Silintan capsules, which is being recalled, should stop using this product and/or return to the place of purchase, discard, or contact their doctor.
Modern Warrior Recalls “Modern Warrior Ready” Dietary Supplement Due to Undeclared 1,4-DMAA and Aniracetam, as Well as Tianeptine, Which has Not Been Approved for Supplement Use by the FDA.
[Phoenix, Arizona] – [12/22/25] – Modern Warrior is voluntarily recalling all lots of Modern Warrior Ready, a dietary supplement sold directly to consumers, after regulatory testing identified the presence of undeclared ingredients, including tianeptine, 1,4-DMAA, and aniracetam.
Tianeptine can cause life-threatening events including suicidal ideation or behavior in children, adolescents, and adults aged 25 years and younger. In addition, overdose carriesserious and potentially life-threatening risks including confusion, seizures, drowsiness, dry mouth, and shortness of breath, which can be exacerbated by alcohol use. Using tianeptine in combination with monoamine oxidase inhibitor (MAOl) antidepressants could lead to life-threatening complications including stroke and death.
Use of 1,4-DMAA can elevate blood pressure and could lead to cardiovascular problems, including heart attack, shortness of breath and tightening of the chest. Products containing synephrine are reported to potentially result in cardiovascular effects, including increasedheart rate and blood pressure, especially in the presence of caffeine.
The recalled product was distributed to customers nationwide from April of 2022 until December 8th of 2025. The recalled product was available for purchase online.
The recalled product is sold in a black plastic supplement bottle with a black screw-top lid sealed with black and gold tamper-evident shrink wrap. The front label features the Modern Warrior (MW) logo in gold at the top.
The product name “Body Repair Plan” appears centered on the front label in gold lettering. Below the product name is the word “READY” with a small sunrise icon, followed by the phrase “Mental Clarity.” The bottle indicates a quantity of 60 capsules.
The overall label design is minimalist, using black and gold coloring with no images other than the brand logo and iconography. The product was sold as a capsule-based dietary supplement under the Modern Warrior brand.
StuffbyNainax LLC Issues Voluntary Nationwide Recall of MR.7 SUPER 700000 Dietary Supplement Due to the Presence of Undeclared Sildenafil and Tadalafil
Huntsville, Texas — StuffbyNainax LLC is voluntarily recalling all lots of MR.7 SUPER 700000 capsules to the consumer level. FDA analysis has found the product to be tainted with sildenafil and tadalafil, which are active ingredients in FDA-approved prescription drugs used to treat male erectile dysfunction and belong to a class of drugs known as phosphodiesterase type‑5 (PDE‑5) inhibitors. Products containing sildenafil or tadalafil cannot be marketed as dietary supplements.
MR.7 SUPER 700000 capsules are considered an unapproved new drug for which safety and efficacy have not been established and are therefore subject to recall.
Risk Statement:
Consumption of products containing undeclared sildenafil or tadalafil may cause serious health risks. These ingredients may interact with nitrates found in certain prescription medications (such as nitroglycerin) and may result in a dangerous drop in blood pressure, which can be life‑threatening. Individuals with diabetes, high blood pressure, high cholesterol, or heart disease frequently take nitrates. Adult males who use nitrates for cardiac conditions are at the greatest risk.
Product Description:
The tainted MR.7 SUPER 700000 capsules are marketed as a dietary supplement for male enhancement. The affected product does not have lot numbers or expiration dates. The product was distributed nationwide in the United States to a limited number of online customers between August 2025 and November 2025.
Affected Product:
Product Name: MR.7 SUPER 700000
Brand: MR.7
Form: Capsules
Lot Number: Not available / unlisted
Distribution Period: August 2025 – November 2025
Distribution: Sold online in the United States
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
WARNING LETTER: Blue Forest Farms, LLC MARCS-CMS 711990 — September 11, 2025
Dear Ms. Johnson:
This is to advise you that the Food and Drug Administration (FDA) reviewed your website at the Internet address www.blueforestfarms.com in July 2025 and has determined that you take orders there for various human food products, which you represent as containing Amanita muscaria. FDA has determined that your products, Amanita Muscaria Microdose Capsules and Amanita Muscaria Magic Potion Tincture, are adulterated under section 402(f)(1)(B) of the Federal Food, Drug, and Cosmetic Act (the Act), 21 U.S.C. 342(f)(1)(B). Additionally, your Amanita Muscaria Magic Chocolate is adulterated under section 402(a)(2)(C)(i) (21 U.S.C. 342(a)(2)(C)(i)) of the Act. As explained further below, introducing or delivering these products for introduction into interstate commerce violates the Act. You can find the Act and FDA regulations through links on FDA’s home page at www.fda.gov.
Adulterated Dietary Supplements
The labeling of your Amanita Muscaria Microdose Capsules and Amanita Muscaria Magic Potion Tincture declares Amanita muscaria as a dietary ingredient. More specifically, the labeling on your Amanita Muscaria Microdose Capsules states that the Amanita muscaria is from whole dry fruiting bodies, and the labeling on your Amanita Muscaria Magic Potion Tincture states that the Amanita muscaria is an extract made from whole dry Amanita Muscaria fruiting bodies. Based on a review of your product labeling, the Amanita muscaria in your Amanita Muscaria Microdose Capsules, is a “dietary ingredient” as a botanical under section 201(ff)(1)(C) of the Act (21.U.S.C. 321(ff)(1)(C)), while the Amanita muscaria in your Amanita Muscaria Magic Potion Tincture is a “dietary ingredient” as an extract of a botanical under section 201(ff)(1)(F) of the Act (21 U.S.C. 321(ff)(1)(F)). Amanita muscaria, A. muscaria extract, ibotenic acid, muscarine, and muscimol are new dietary ingredients under section 413(d) of the Act (21 U.S.C. 350b(d)) because, to the best of FDA’s knowledge, there is no information demonstrating that Amanita muscaria was marketed as a dietary ingredient in the United States before October 15, 1994.
A dietary supplement is adulterated under section 402(f)(1)(B) of the Act (21 U.S.C. 342(f)(1)(B)) if it contains a new dietary ingredient for which there is inadequate information to provide reasonable assurance that such ingredient does not present a significant or unreasonable risk of illness or injury. Based on our evaluation of the relevant safety evidence, there is inadequate information to provide reasonable assurance that Amanita muscaria does not present a significant or unreasonable risk of illness or injury. Therefore, your dietary supplements containing Amanita muscaria are adulterated under section 402(f)(1)(B) of the Act. The introduction of such adulterated dietary supplements into interstate commerce is prohibited under section 301(a) of the Act. Moreover, available information on the use of Amanita muscaria in products raises serious safety concerns.
Adulterated Human Foods
Based on a review of your product labeling, your Amanita Muscaria Magic Chocolate is a conventional food to which Amanita muscaria has been added.
As defined in section 201(s) of the Act (21 U.S.C. 321(s)), the term “food additive” refers to any substance the intended use of which results in it becoming a component of any food, unless the substance is generally recognized as safe (GRAS) among qualified experts under the conditions of its intended use, or unless the substance meets a listed exception.1
Additionally, your Amanita Muscaria Magic Chocolate product is represented for use as a conventional food, and accordingly is not a dietary supplement as labeled, and as defined under Section 201(ff) of the Act (21 U.S.C. 321(ff)). The Act excludes from the definition of a dietary supplement a product represented for use as a conventional food or as a sole item of a meal or the diet (21 U.S.C. 321(ff)(2)(B)). Your use of “Dietary Supplement” as a statement of identity and a “Supplement Facts” panel for nutrition labeling does not make your product a dietary supplement because your Amanita Muscaria Magic Chocolate product is represented for use as a conventional food. Examples of factors and information that establish that the product is represented as a conventional food follow:
Chocolate Bar:
The use of a combination of ingredients characteristic to a chocolate bar which are listed on the website (including chocolate, sugar, cocoa butter, soy lecithin, natural vanilla flavor, and milk); and
The appearance and packaging of the product as a chocolate bar.
As sold, your Amanita Muscaria Magic Chocolate product is represented for use as a conventional food, for example, by the use of the word “Chocolate” in the statement of identity on the package label.
Food additives require premarket approval based on data demonstrating safety. Any food additive that has not been approved for its intended use in food is deemed to be unsafe under section 409(a) of the Act (21 U.S.C. 348(a)) and causes the food to be adulterated under section 402(a)(2)(C)(i) of the Act, 21 U.S.C. 342(a)(2)(C)(i). Introduction of an adulterated food into interstate commerce is prohibited under section 301(a) of the Act, 21 U.S.C. 331(a).
There is no food additive regulation that authorizes the use of Amanita muscaria. We are not aware of any information to indicate that Amanita muscaria is the subject of a prior sanction (see 21 CFR Part 181). Furthermore, we are not aware of any basis to conclude that Amanita muscaria is GRAS for use in conventional foods. FDA’s regulations in 21 CFR 170.30(a)-(c) describe the criteria for eligibility for classification of a food ingredient as GRAS. The use of a food substance may be GRAS based on either scientific procedures or, for a substance used in food before 1958, through experience based on common use in food (see 21 CFR 170.30).
We know of no basis for general recognition of safety for Amanita muscaria based either on scientific procedures or common use in food prior to January 1, 1958. Based on our review of published scientific literature, existing data and information do not provide an adequate basis to conclude that the use of Amanita muscaria in food meets the criteria for GRAS status. Some of the available data raise serious concerns about potential harm associated with Amanita muscaria. In our review, we identified published scientific literature, adverse event reports, and calls to poison control centers that reported serious adverse effects on the central nervous system associated with consumption of Amanita muscaria, including delirium, seizures, coma, respiratory depression, and potentially death. Therefore, based on our review, the use of Amanita muscaria in conventional food does not satisfy the criteria for GRAS status under 21 CFR 170.30.
FDA is not aware of any other exception to the food additive definition that would apply to Amanita muscaria for use as an ingredient in a conventional food. Therefore, Amanita muscaria when added to a conventional food is a food additive under section 201(s) of the Act and is subject to the provisions of section 409 of the Act. Under section 409, a food additive is deemed unsafe unless it is approved by FDA for its intended use prior to marketing. Amanita muscaria is not approved for use in any conventional food. Food containing an unsafe food additive within the meaning of section 409 is adulterated within the meaning of section 402(a)(2)(C)(i) of the Act. Therefore, your Amanita Muscaria Magic Chocolate, and other foods that contain Amanita muscaria, are adulterated within the meaning of section 402(a)(2)(C)(i) of the Act because they bear or contain an unsafe food additive. The introduction of these adulterated foods into interstate commerce is prohibited under section 301(a) of the Act, 21 U.S.C. 331(a).
Source: https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/blue-forest-farms-llc-711990-09112025
One Source Nutrition, Inc. Issues Voluntary Nationwide Recall of Vitality Capsules Due to Presence of Undeclared Sildenafil and Tadalafil
Benton, Arkansas – 02/2025 –One Source Nutrition is voluntarily recalling all lots of Vitality capsules to the Consumer level. FDA analysis has found the product to be tainted with sildenafil and tadalafil, which are ingredients in FDA approved products for treatment of male erectile dysfunction in the family of drugs known as phosphodiesterase (PDE-5) inhibitors. Products containing sildenafil and tadalafil cannot be marketed as dietary supplements. Vitality capsules is an unapproved new drug for which safety and efficacy have not been established and, therefore, subject to recall.
Risk Statement: Consumption of products with undeclared tadalafil may interact with nitrates found in some prescription drugs (such as nitroglycerin) and may cause a significant drop in blood pressure that may be life threatening. People with diabetes, high blood pressure, high cholesterol, or heart disease often take nitrates. Among the adult male population, who are most likely to use this product, adult males who use nitrates for cardiac conditions are most at risk. To date, One Source Nutrition, Inc. has not received any reports of adverse events related to this recall.
The tainted Vitality capsules is marketed as a dietary supplement used as a male enhancement product and is packaged in a single pill. The affected Vitality capsules product does not have lot numbers or expiration dates. The product can be identified by a single pill in an orange and gray package with blue writing. Vitality capsules was distributed Nationwide to retail outlets from wholesale distributor.
Shoppers-Plaza Issues Voluntary Nationwide Recall of Fouzee Sugarlin Herbal Formula Due To Presence Of Undeclared Metformin And Glyburide
December 16, 2024 – Los Angeles, California – Shoppers-Plaza is voluntarily recalling all lots of Fouzee Sugarlin Herbal Formula capsules to the consumer level. See Attachment 1 for photo of product labeling. Fouzee Sugarlin Herbal Formula capsules is an unapproved new drug for which safety and efficacy have not been established and, therefore, subject to recall.
Metformin is the active ingredient in several FDA-approved prescription drugs used to treat type 2 diabetes and is only available with a prescription. Metformin may pose a serious risk to consumers because this ingredient can cause a rare but serious side effect called lactic acidosis (a build-up of lactic acid in the blood) that can cause death if untreated. Symptoms of lactic acidosis include nausea, vomiting, fatigue, body aches, respiratory distress, abdominal pain and fast, deep breathing. Metformin may also cause hypoglycemia when taken together with other medications to lower blood sugar.
Glyburide is the active ingredient in some FDA-approved prescription drugs used to treat type 2 diabetes and is only available with a prescription. Glyburide may pose serious and potentially life-threatening risks to consumers because this ingredient may result in unsafe drops in blood sugar levels (hypoglycemia). Common symptoms of hypoglycemia include fast heartbeat, shaking, sweating, nervousness or anxiety, irritability or confusion, dizziness, drowsiness, headache and hunger. Hypoglycemia may quickly become severe, resulting in seizures, coma or death.
This product was promoted and sold nationwide as a glucose or blood sugar support supplement on various websites including, www.shoppers-plaza.comExternal Link Disclaimer and possibly in some retail stores.
The recalled Fouzee SugarLin Herbal Formula is packaged in a white bottle containing 180 veg capsules, with UPC 26656690477.
Source: https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/shoppers-plaza-issues-voluntary-nationwide-recall-fouzee-sugarlin-herbal-formula-due-presence
GNMART INC Issues Voluntary Nationwide Recall of Force Forever Due to Undeclared Drug Ingredients Diclofenac and Dexamethasone
12/12/2024, Dover Plains, NY, GNMART INC is voluntarily recalling all lots of Force Forever for joint Pain, 60 Tablets packaged in a white plastic bottle with a red cap, labeled as “FORCE FOREVER.”, to the consumer level. FDA analysis has found product to contain undeclared Diclofenac and Dexamethasone.
Risk Statement: Diclofenac is a non-steroidal anti-inflammatory drug (commonly referred to as NSAIDs). NSAIDs may cause increased risk of cardiovascular events, such as heart attack and stroke, as well as serious gastrointestinal damage, including bleeding, ulceration, and fatal perforation of the stomach and intestines. Dexamethasone is a corticosteroid commonly used to treat inflammatory conditions. Corticosteroid use can impair a person’s ability to fight infections and can cause high blood sugar levels, muscle injuries and psychiatric problems. GNMart has not received any reports of adverse events related to this recall.
The product is used as a dietary supplement for joint pain and is packaged in bottles with 60 tablets. The affected product includes all lots and expiration date: 03/27/2030. The product was distributed nationwide via the internet at gnmart.comExternal Link Disclaimer.
FDA Alerts Industry and Consumers about the Use of Amanita Muscaria or its Constituents in Food
December 18, 2024
Today, the U.S. Food and Drug Administration (FDA) issued a letter to food manufacturers that Amanita muscaria (A. muscaria), its extracts, and certain of its constituents (muscimol, ibotenic acid, and muscarine) are not authorized for use as ingredients in conventional food. A. muscaria and its constituents have been used in foods intended to have hallucinogenic effects, sometimes marketed as “psychedelic edibles”, “legal psychedelics” or “mushroom edibles”. After reviewing the available information about A. muscaria and its constituents, the FDA concluded that they do not meet the safety standard for use in food and that their use as food ingredients may be harmful. We also recommend that people avoid eating foods with these ingredients. The FDA’s assessment of chemicals in the food supply is part of our commitment to food safety and public health.
The FDA determined that the use of this ingredient and its constituents do not meet the Generally Recognized As Safe (or GRAS) standard and that they are unapproved food additives. The agency included its assessment in a memorandum added to the Post-market Determinations that the Use of a Substance is Not GRAS inventory. The agency updates this inventory periodically and we encourage food manufacturers to use it as a resource. The FDA reminds industry to engage the agency through the GRAS Notification Program, in which FDA experts evaluate the notifier’s GRAS conclusion which includes the notifier’s analysis of publicly available safety data.
The FDA is aware of these ingredients in foods intended to have hallucinogenic effects that look like their conventional counterparts, like candy bars. Because of the FDA’s safety concerns with these ingredients, the agency reminds manufacturers considering using them in foods of their responsibility to produce and market safe foods. This includes ensuring the safety and regulatory status of the ingredients they use in foods before they are available to consumers.
We chose to assess the use of this ingredient and its constituents in food because of new information about its safety profile, including adverse event reports, news reports, and inquiries from state and local regulators about the regulatory status and safety of this ingredient. This is not the first time we have shared our concerns about A. muscaria. The FDA’s “Bad Bug” book, released in 2012, identifies A. muscaria and its constituents as toxic agents that cause poisoning following ingestion.
As part of a broader assessment of these ingredients in the food supply, the FDA is also evaluating the use of these ingredients in dietary supplements. The FDA reminds manufacturers that are exploring the use of A. muscaria or its constituents in dietary supplements of their responsibility to ensure their ingredients meet the relevant safety standard and encourages them to engage the Office of Dietary Supplement Programs with questions.
Buy-herbal.com Issues Voluntary Nationwide Recall of Nhan Sam Tuyet Lien Truy Phong Hoan Capsules Due to Undeclared Furosemide, Dexamethasone and Chlorpheniramine
12th December 2024, Flushing, New York, Buy-herbal.com is voluntarily recalling all lots within expiry of Nhan Sam Tuyet Lien Truy Phong Hoan Capsules to the consumer level. FDA analysis has found these products to contain undeclared Furosemide, Dexamethasone and Chlorpheniramine. Furosemide was found at 5.24 mg/g or 1.84 mg/capsule. Dexamethasone was found at 2.22 mg/g or 0.780 mg/capsule. Chlorpheniramine was found at 4.38 mg/g or 1.54 mg/capsule. Products containing Furosemide, Dexamethasone and Chlorpheniramine cannot be marketed as dietary supplements. Nhan Sam Tuyet Lien Truy Phong Hoan Capsules is an unapproved drug for which safety and efficacy have not been established and therefore, subject to recall.
Risk Statement: Chlorpheniramine is an antihistamine available in several over the counter products. Inadvertent consumption of chlorpheniramine could lead to a higher than intended dose, which could potentially cause overexcitement, loss of coordination, drowsiness, loss of consciousness, and seizures. Dexamethasone is a corticosteroid commonly used to treat inflammatory conditions. Corticosteroid use can impair a person’s ability to fight infections and can cause high blood sugar levels, muscle injuries and psychiatric problems. When corticosteroids are taken for a prolonged period, or at high doses, they can suppress the adrenal gland. Abrupt discontinuation can cause withdrawal symptoms. In addition, the undeclared dexamethasone may cause serious side effects when combined with other medications. Furosemide is a potent diuretic used to treat conditions such as, congestive heart failure, high blood pressure, and edema. It can cause dizziness, sudden drop in blood pressure when standing up from a seated or lying position, hearing loss, kidney damage, and profound dehydration and electrolyte imbalance. Patients allergic to sulfonamides may also be allergic to furosemide. Buy-herbal.com has not received any reports of adverse events related to this recall.
This is marketed as a dietary supplement for treatment for lumbago gout, arthrodynia, myasthenia, limb numbness, pain induced by acclimation fever and rheumatism, osteocope and arthritis. The product is packaged in bottle containing 30 capsules each. The affected Nhan Sam Tuyet Lien Truy Phong Hoan include all batches within Expiration dates.
C&A Naturistics Issues Voluntary Nationwide Recall of AK Forte Tablets con Ortiga y Omega 3 Due to the Presence of Undeclared Drug Ingredients: Diclofenac, Dexamethasone, and Methocarbamol
10/8/24 – National City, CA, C&A Naturistics is voluntarily recalling all lots of AK Forte, 400 mg tablets, to the consumer level. FDA analysis has found the product to be tainted Diclofenac, Dexamethasone, and Methocarbamol. Products containing diclofenac, dexamethasone, and methocarbamol cannot be marketed as dietary supplements. AK Forte tablets is an unapproved new drug for which safety and efficacy have not been established and, therefore, subject to recall.
Risk Statement: Dexamethasone is a corticosteroid commonly used to treat inflammatory conditions. Corticosteroid use can impair a person’s ability to fight infections and can cause high blood sugar levels, muscle injuries and psychiatric problems. When corticosteroids are taken for a prolonged period, or at high doses, they can suppress the adrenal gland. Abrupt discontinuation can cause withdrawal symptoms. In addition, the undeclared dexamethasone in AK Forte may cause serious side effects when combined with other medications. Diclofenac is a non-steroidal anti-inflammatory drug (commonly referred to as NSAIDs). NSAIDs may cause increased risk of cardiovascular events, such as heart attack and stroke, as well as serious gastrointestinal damage, including bleeding, ulceration, and fatal perforation of the stomach and intestines. This hidden drug ingredient may also interact with other medications and significantly increase the risk of adverse events, particularly when consumers use multiple NSAID-containing products. Methocarbamol is a muscle relaxant that can cause sedation, dizziness, and low blood pressure. Methocarbamol can also impair mental and physical abilities to perform certain tasks, such as driving a motor vehicle or operating machinery. To date, C&A Naturistics has not received any reports of adverse events related to this recall.
AK Forte is marketed as a dietary supplement for Joint pain and Arthritis and is packaged in a cardbox type of packaging, with white, blue, red and gold logo of AK with a gold crown on top with 100 tablets per units, and all codes. The affected product AK Forte, all lots were distributed Nationwide via our Ebay and Etsy store.
Source: https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/ca-naturistics-issues-voluntary-nationwide-recall-ak-forte-tablets-con-ortiga-y-omega-3-due-presence
123Herbals LLC (123Herbals.com) Issues Voluntary Nationwide Recall of Vail-Bon Jie Yang Wan Capsules Due to the Presence of Undeclared Dexamethasone and Chlorpheniramine
09/18/2024 – Rosemead, CA, 123herbals is voluntarily recalling all lots of Vail-Bon Jie Yang Wan capsules to the consumer level. The product has been found to be tainted with the drug ingredients, dexamethasone and chlorpheniramine. Vail Bon Jie Yang Wan capsules is an unapproved new drug for which the safety and efficacy have not been established and, therefore, subject to recall.
Risk Statement: If used chronically at the recommended dose, dexamethasone could cause severe and serious adverse events such as adrenal suppression (a disorder in which the adrenal glands do not produce enough hormones), central nervous system and psychiatric/behavioral effects, weight gain, gastrointestinal effects, elevated blood glucose, increased infection risks, neuromuscular and skeletal side effects, ocular effects, cardiovascular effects, dermatologic effects endocrine and metabolic issues, among other adverse events not mentioned. Depending on the dose and duration of the systemic dexamethasone, these adverse events may be severe and even life threatening. Chlorpheniramine is an antihistamine available in several over the counter products. Inadvertent use of tainted Vail-Bon Jie Yang Wan capsules could lead to a higher than intended chlorpheniramine dose, which could potentially cause overexcitement, loss of coordination, drowsiness, loss of consciousness, and seizures. To date, 123Herbals has not received any reports of adverse events related to this recall.
The product is used to treat skin disease, eczema, itch and is packaged in 30-count plastic bottles. The affected Vail-Bon Jie Yang Wan capsules include all lot numbers and expirations. Vail- Bon Jie Yang Wan capsules were distributed nationwide on 123herbals.comExternal Link Disclaimer and online retailers including Amazon.
GURU INC. Issues Voluntary Nationwide Recall of Infla-650 Herbal Dietary Supplement Capsules Due to Hidden Drug Ingredients
07/16/2024 – Lilburn, GA, GURU INC. is voluntarily recalling Infla-650 Herbal Dietary Supplement Capsules, Lot No. IN-032, 700 mg to the consumer/user level. Infla-650 capsules has been found to be tainted with acetaminophen, diclofenac and phenylbutazone, which are not listed on the label. Products containing acetaminophen, diclofenac and phenylbutazone cannot be marketed as dietary supplements. Infla-650 Herbal Dietary Supplement Capsules is an unapproved new drug for which safety and efficacy have not been established and, therefore, subject to recall.
Risk Statement: Use of the product poses a serious threat to consumers because it may result in unintentional acetaminophen overdose, especially if it is used in combination with other acetaminophen-containing products. Acetaminophen overdose can cause liver damage (hepatoxicity), ranging in severity from abnormalities in liver function to acute liver failure, and even death. People who experience unintentional acetaminophen overdose may not seek prompt medical attention because the symptoms of liver damage can take several days to emerge, even in severe cases. Acetaminophen may also cause serious skin reactions. Symptoms may include skin reddening, rash, and blisters.
Diclofenac is a non-steroidal anti-inflammatory drug (commonly referred to as NSAIDs). NSAIDs may cause increased risk of cardiovascular events, such as heart attack and stroke, as well as serious gastrointestinal damage, including bleeding, ulceration, and fatal perforation of the stomach and intestines. This hidden drug ingredient may also interact with other medications and significantly increase the risk of adverse events, particularly when consumers use multiple NSAID-containing products
Phenylbutazone is a NSAID that was discontinued for human use in the United States due to the risk of serious and life-threatening injuries. The most serious and life-threatening injury associated with phenylbutazone treatment is bone marrow toxicity, which occurs when the body does not produce enough red blood cells, white blood cells, and/or platelets. Certain types of bone marrow toxicity are reversible; however, in rare circumstances it can lead to death. Patients with anemia, low white or red blood cell count, or decreased blood platelets may be at a greater risk of death or serious injury.
Source: https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/guru-inc-issues-voluntary-nationwide-recall-infla-650-herbal-dietary-supplement-capsules-due-hidden
Supercore Products Group, Inc. Issues Voluntary Worldwide Recall of Hard Steel Capsules & Gold Hard Steel Plus Liquid Due to the Presence of Sildenafil and Acetaminophen
7/10/2024 – Atlanta, GA, Supercore Products Group is voluntarily recalling of Hard Steel Capsules & Gold Hard Steel Plus Liquid to the consumer level. FDA analysis has found these products to be tainted with Sildenafil and acetaminophen. Sildenafil is an ingredient known as a Phosphodiesterase Inhibitor (PDE-5) found in FDA approved products for the treatment of male erectile dysfunction. The presence of sildenafil in these products renders them an unapproved drug for which the safety and efficacy has not been established and, therefore, subject to recall.
Risk Statement: Consumption of products with undeclared sildenafil may interact with nitrates found in some prescription drugs (such as nitroglycerin) and may cause a significant drop in blood pressure that may be life threatening. People with diabetes, high blood pressure, high cholesterol, or heart disease often take nitrates. Among the adult male population, who are most likely to use this product, those who use nitrates for cardiac conditions are most at risk.
Acetaminophen is a medicine used to reduced pain and fever that is found in many prescriptions, and over-the-counter (OTC) drug products. Use of the products poses a serious threat to consumers because it may result in unintentional acetaminophen overdose, especially if it is used in combination with other acetaminophen-containing products. Acetaminophen overdose can cause liver damage (hepatoxicity), ranging in severity from abnormalities in liver function to acute liver failure, and even death. Victims of unintentional acetaminophen overdose may not seek prompt medical attention because the symptoms of liver damage can take several days to emerge, even in severe cases. Acetaminophen may also cause serious skin reactions. Symptoms may include skin reddening, rash, and blisters.
As of to date, Supercore Products Group has not received any reports of adverse events related to this recall.
The recalled products are used as a male enhancement sold at https://hardsteelusa.com and are sold in single, 10ct, 20ct and 30ct packages. The affected Hard Steel Capsules include the following purchase period: January 5th, 2024 through July 5th, 2024. The product can be identified by its original packaging and black/white capsule or black/gold capsule. Gold Hard Steel Plus Liquids are sold in 2oz bottles. This product is sometime described as Hard Steel Gold 2oz Liquid Shots, packaged in black and gold bottles.Source: https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/supercore-products-group-inc-issues-voluntary-worldwide-recall-hard-steel-capsules-gold-hard-steel
June 11, 2024: Integrity Products , St Louis MO is voluntarily recalling tainted lot (HGW221116) within expiry of the Ram It & To The Moon capsules to the consumer level.
FDA analysis founds these products to be tainted with sildenafil and Tadalafil. Sildenafil/Tadalafil is an FDA approved drug for the treatment of erectile dysfunction, the presence of sildenafil in the Ram It & To The Moon products renders them unapproved drugs for which safety and efficacy have not been established, therefor subject to recall.
Risk Statements: Men with diabetes, high blood pressure, high cholesterol, or heart disease, may be on medications that if taken with these products could lower blood pressure to dangerous levels that could be life threatening. The products affected are men with diabetes, high blood pressure, high cholesterol, or heart disease. To date, Integrity Products. has not received any reports of adverse events related to this recall.
The product is marketed as dietary supplements for male sexual enhancement and is packaged in a blister card. 10 count boxes. Ram It & To The Moon were distributed to consumers nationwide via the Internet. We are notifying the public through this public announcement due to lack of ability to identify customers who may have received the product. Integrity Products. is notifying its customers that have the Ram It & To The Moon products to stop use and properly discard the product.
04/22/2024– Hollywood, FL, US, STOP CLOPEZ CORP is voluntarily recalling one lot of Schwinnng capsules to the consumer level.
FDA analysis has found the Schwinnng products to contain Nortadalafil. Nortadalafil is an active drug ingredient known for the treatment of male erectile dysfunction. The presence of Nortadalafil in Schwinnng capsules makes it an unapproved new drug for which the safety and efficacy have not been established and, therefore subject to recall.
Risk Statement: The Schwinnng products contain Nortadalafil, which may interact with nitrates found in some prescription drugs, such as nitroglycerin, and may cause a significant drop in blood pressure that may be life threatening. People with diabetes, high blood pressure, high cholesterol, or heart disease often take nitrates. Among the adult male population, who are most likely to use this product, adult males who use nitrates for cardiac conditions are most at risk. To date, no adverse events have been reported.
This product is marketed as a dietary supplement for male sexual enhancement and is packaged and coded as follows:
SCHWINNNG Lot 2108 EXP 10/2024The product was distributed Worldwide via Amazon at www.amazon.com
Pyramid Wholesale Issues Recall of Various Brands of Products Sold as Dietary Supplements for Sexual Enhancement Because They Contain Undeclared Prescription Drugs Including Sildenafil (Viagra) and/or Tadalafil (Cialis)
March 19, 2024, Los Angeles, CA – Pyramid Wholesale is issuing a recall of various brands of products sold as dietary supplements for sexual enhancement because they contain undeclared prescription drugs including Sildenafil (Viagra) and/or Tadalafil (Cialis).
These products have the potential to cause severe adverse health effects due to misuse, overuse, interaction with other medications, underlying health conditions, and overall lack of oversight or consultation with medical professionals
The products were distributed under 11 different brand names and varied in product form and size. They were sold in bulk to other distributors and retailers in California and out of state. A full list is available below.
There have been no reports of injury or illness, to date.
Consumers should immediately discontinue use of the product.Source: https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/pyramid-wholesale-issues-recall-various-brands-products-sold-dietary-supplements-sexual-enhancement
WARNING LETTER: Carbon Fire, LLC MARCS-CMS 664700 — March 01, 2024
Dear David Woods:
This letter concerns your product 213° Metabolism Boosting Complex, which is labeled as a dietary supplement. Your product labeling declares N-Methyltyramine as a dietary ingredient in 213° Metabolism Boosting Complex. As explained further below, introducing or delivering this product for introduction into interstate commerce violates the Federal Food, Drug, and Cosmetic Act (the Act) because the product is an adulterated dietary supplement. You can find the Act and FDA regulations through links on FDA’s home page at www.fda.gov.
The term “dietary supplement” is defined in section 201(ff) of the Act [21 U.S.C. § 321(ff)]. NMethyltyramine is a “dietary ingredient” under section 201(ff)(1)(F) of the Act because it is a constituent of various botanicals. N-Methyltyramine is also a “new dietary ingredient” (i.e., a dietary ingredient not marketed in the United States before October 15, 1994) under section 413(d) of the Act [21 U.S.C. § 350b(d)].
Under section 413(a) of the Act [21 U.S.C. § 350b(a)], a dietary supplement that contains a new dietary ingredient shall be deemed adulterated under section 402(f) of the Act [21 U.S.C. § 342(f)] unless it meets one of two requirements:
1. The dietary supplement contains only dietary ingredients that have been present in the food supply as an article used for food in a form in which the food has not been chemically altered; or
2. There is a history of use or other evidence of safety establishing that the dietary ingredient when used under the conditions recommended or suggested in the labeling of the dietary supplement will reasonably be expected to be safe and, at least 75 days before being introduced or delivered for introduction into interstate commerce, the manufacturer or distributor of the dietary ingredient or dietary supplement provides FDA with information, including any citation to published articles, which is the basis on which the manufacturer or distributor has concluded that a dietary supplement containing such dietary ingredient will reasonably be expected to be safe.
To the best of FDA’s knowledge, there is no information demonstrating that N-Methyltyramine was lawfully marketed as a dietary ingredient in the United States before October 15, 1994, nor is there information demonstrating that this ingredient has been present in the food supply as an article used for human food in a form in which the food has not been chemically altered. As such, N-Methyltyramine is subject to the notification requirement in section 413(a)(2) of the Act [21 U.S.C. § 350b(a)(2)] and 21 CFR 190.6. FDA has not received any new dietary ingredient notifications pertaining to the use of NMethyltyramine in dietary supplements. Products for which the manufacturer or distributor is required to submit a new dietary ingredient notification under section 413(a)(2) and 21 CFR 190.6, but for which the required notification has not been submitted, are adulterated under sections 402(f) and 413(a) of the Act [21 U.S.C. §§ 342(f) and 350b(a)]. Introduction of such adulterated dietaty supplements into interstate commerce is prohibited under section 301(a) and (v) of the Act [21 U.S.C. § 331(a) and (v)].
Source: https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/carbon-fire-llc-664700-03012024
Neptune Resources, LLC Issues Voluntary Nationwide Recall of Neptune’s Fix
Jan. 28, 2024 /PRNewswire-PRWeb/ — Neptune Resources, LLC is voluntarily recalling All lots of Neptune’s Fix Elixir, Neptune’s Fix Extra Strength Elixir, and Neptune’s Fix Tablets to the consumer level. Neptune Resources LLC’s distribution channels have not reported any adverse events from the use of its products. The products are being recalled because they contain tianeptine, an ingredient that is not FDA-approved for any medical use. The presence of tianeptine renders the products unapproved drugs for which safety and efficacy have not been established and, therefore, are subject to recall.
Risk Statement: The FDA has advised that: There is a reasonable probability of life-threatening events including suicidal ideation or behavior for children, adolescents, and young adults 25 and younger. In addition, individuals could unintentionally overdose and experience serious and potentially life-threatening risks including confusion, seizures, drowsiness, dry mouth, and shortness of breath, which may be exacerbated by alcohol use. Further, the risks of adverse effects associated with use of tianeptine along with antidepressants, known as monoamine oxidase inhibitors (MAOls), are potentially serious and life-threatening.
Neptune’s Fix Elixir and Extra Strength Elixir are used as a supplement and are packaged in amber glass bottles with a “shrink sleeve” label that covers the entire cap/bottle and is perforated at the cap to facilitate opening. Neptune’s Fix tablets are packaged in 20-count blister packs held in small boxes or 4-count foil packets. All Neptune’s Fix products are being recalled. The product can be identified by the name Neptune’s Fix and its logo which is an illustration of the Roman God Neptune with a green beard and a trident. The products were distributed Nationwide to wholesale and retail customers.Source: https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/neptune-resources-llc-issues-voluntary-nationwide-recall-neptunes-fix
Defyned Brands / 5 Star Nutrition LLC (January 12, 2024)
Defyned Brands, an Austin, Texas, company also known as 5 Star Nutrition LLC, pleaded guilty today to a three-count information charging it with distributing misbranded dietary supplements.
Pursuant to the plea agreement, the company admitted that from September 2018 to July 2020, it delivered into interstate commerce misbranded dietary supplements, which are considered a type of food under the federal Food, Drug and Cosmetic Act (FDCA). The company specifically admitted that shipments of products known as Epivar, Alpha Shredded and Laxobolic were misbranded. According to the plea agreement, the products contained ingredients mislabeled as dietary ingredients or not listed on the product label.
The products at issue were marketed as workout supplements and sold at 5 Star Nutrition retail locations. As part of the plea, the company agreed to forfeit $4.5 million and comply with the terms of a compliance program and certain compliance reporting requirements. Magistrate Judge Susan Hightower of the U.S. District Court for the Western District of Texas presided over the plea.
Source: https://www.justice.gov/opa/pr/texas-company-pleads-guilty-distributing-misbranded-dietary-supplements-and-agrees-45
Amazon.com (December 20, 2023)
FDA WARNING LETTER EXCERPT
Andy Jassy, CEO
Amazon.com, Inc.
2021 7th Ave
Seattle, WA 98121-2601
RE: 662503
Dear Mr. Jassy:
This letter concerns your firm’s distribution of products that violate the Federal Food, Drug, and Cosmetic Act (the “FD&C Act”). The United States Food and Drug Administration (FDA) purchased on your website, www.amazon.com, products that are labeled as energy enhancing supplements or food, but laboratory analyses confirmed that they contained undeclared and potentially harmful active pharmaceutical ingredients…
FDA purchased “MANNERS Energy Boost,” “Round 2,” “WeFun,” “Genergy,” “Big Guys Male Energy Supplement,” “Mens Maximum Energy Supplement,” and “X Max Triple Shot Energy Honey” through your website, www.amazon.com. All of these products were introduced or delivered for introduction into interstate commerce by Amazon via your Fulfillment by Amazon service.1 FDA confirmed through laboratory analyses that the “MANNERS Energy Boost,” “Round 2,” “Genergy,” and “X Max Triple Shot Energy Honey” products, purchased on www.amazon.com, contained the active pharmaceutical ingredient (API) tadalafil; and the “WeFun,”2 “Big Guys Male Energy Supplement,” and “Mens Maximum Energy Supplement” products, also purchased on www.amazon.com, contained the API sildenafil. These ingredients are not declared on the products’ labeling.3 Sildenafil and tadalafil are phosphodiesterase type-5 (PDE-5) inhibitors and the active ingredients in the FDA-approved prescription drugs Viagra and Cialis, respectively, used to treat erectile dysfunction (ED). These undeclared ingredients may interact with nitrates found in some prescription drugs, such as nitroglycerin, and may lower blood pressure to dangerous levels.
Source: https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/amazoncom-inc-662503-12202023
Hua Da Trading (December 20, 2023)
FDA WARNING LETTER EXCERPT
Daniel Zeng
Hua Da Trading, Inc.
PO Box 40517
Brooklyn, NY 11204
RE: 664359
Dear Daniel Zeng:
This letter is to advise you that the United States Food and Drug Administration (FDA) reviewed your website at the Internet address, https://www.eshoponlineusa.com, from July to December 2023, where you take orders for “WeFun.”1 In addition, FDA has obtained a sample and labeling of your “WeFun” product. As described below, this product is an unapproved new drug sold in violation of sections 505(a) and 301(d) of the Federal Food, Drug, and Cosmetic Act (FD&C Act), 21 U.S.C. 355(a) and 331(d). Furthermore, your “WeFun” is a misbranded drug under section 502 of the FD&C Act, 21 U.S.C. 352, and sold in violation of section 301(a) of the FD&C Act, 21 U.S.C. 331(a).
FDA confirmed through laboratory analysis that a sample of your “WeFun” product contains the undeclared pharmaceutical ingredient sildenafil. Sildenafil is a phosphodiesterase type-5 (PDE-5) inhibitor and the active ingredient in the FDA-approved prescription drug Viagra used to treat erectile dysfunction (ED). This undeclared ingredient may interact with nitrates found in some prescription drugs, such as nitroglycerin, and may lower blood pressure to dangerous levels. Men with diabetes, high blood pressure, high cholesterol, or heart disease, often take nitrates.
Source: https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/hua-da-trading-inc-664359-12202023
Artri King / Botanical Be Warning Letter (December 20, 2023)
Edgar H. Felix
Botanical Be
360 Jardin Bello
El Paso, TX 79932-2357
RE: 671066
Dear Mr. Felix:
This letter is to advise you that the United States Food and Drug Administration (FDA) reviewed your website at the Internet address https://botanical-be.com/ in July 2023, where you take orders for “Kuka Flex Forte,” “Reumo Flex,” and “Artri King Reforzado con Ortiga y Omega 3” products. In addition, FDA has obtained samples and labeling of your “Kuka Flex Forte,” “Reumo Flex,” and “Artri King Reforzado con Ortiga y Omega 3” products. As described below, these products are unapproved new drugs sold in violation of sections 505(a) and 301(d) of the Federal Food, Drug, and Cosmetic Act (FD&C Act), 21 U.S.C. 355(a) and 331(d). Furthermore, the products are misbranded drugs under section 502 of the FD&C Act, 21 U.S.C. 352 and sold in violation of section 301(a) of the FD&C Act, 21 U.S.C. 331(a).
FDA confirmed through laboratory analysis that a sample of your “Kuka Flex Forte,” “Reumo Flex,” and “Artri King Reforzado con Ortiga y Omega 3” contains undeclared diclofenac. Diclofenac is a non-steroidal anti-inflammatory drug (commonly referred to as NSAIDs). NSAIDs may cause increased risk of cardiovascular events, such as heart attack and stroke, as well as serious gastrointestinal damage, including bleeding, ulceration, and fatal perforation of the stomach and intestines. This hidden drug ingredient may also interact with other medications and significantly increase the risk of adverse events, particularly when consumers use multiple NSAID-containing products.
Total Body Nutrition LLC, TBN Labs LLC, and Loud Muscle Science, LLC, Mohammed Islam (December 18, 2023)
A civil complaint filed on October 18, 2023 at the request of the U.S. Food and Drug Administration (FDA), alleged that Total Body Nutrition LLC, TBN Labs LLC, and Loud Muscle Science, LLC (collectively, “TBN companies”), and the companies’ owner, Mohammed Islam, violated the FDCA at the companies’ facility in Hauppauge, Long Island, and their previous facility in Edgewood, New York, by manufacturing and distributing adulterated and misbranded dietary supplements. The complaint alleges that Islam and the TBN companies violated the FDCA by manufacturing dietary supplements without establishing product specifications for the finished batches and without testing or examining the finished batches to verify that they met product specifications, and by using dietary ingredients in their dietary supplements without first testing or examining the ingredients to verify their identity. The complaint also alleged that FDA inspected the TBN companies’ current and previous facilities four times, in 2017, 2018, 2021, and 2023, and found violations of the FDCA at each inspection. According to the complaint, FDA also issued Islam and the TBN companies warning letters in 2016, 2017, and 2019.
Islam and the TBN companies agreed to settle the suit and be bound by a consent decree of permanent injunction. The negotiated consent decree entered by the court enjoins Islam and the TBN companies from violating the FDCA, and requires, among other things, that Islam and the TBN companies comply with the dietary supplement current good manufacturing practice regulations and the dietary supplement labeling provisions of the FDCA and its implementing regulations. Further, Islam and the TBN companies must destroy all of their adulterated dietary supplements.
Balance of Nature and Evig LLC, and Ryan Petersen (November 16, 2023)
In a complaint filed on Oct. 11 in the U.S. District Court for the District of Utah at the request of the U.S. Food and Drug Administration, the United States alleged that Evig LLC and the company’s CEO, David Lex Howard, violated the federal Food, Drug and Cosmetic Act (FDCA) by distributing adulterated and misbranded dietary supplements. In a separate complaint filed the same day, the United States alleged that Premium Productions LLC and the company manager’s, Ryan Petersen, violated the FDCA by manufacturing adulterated dietary supplements. According to the complaints, the dietary supplements involved are marketed throughout the United States under the brand name Balance of Nature.
The U.S. District Court for the District of Utah, Central Division has entered two consent decrees of permanent injunction against Evig LLC, of St. George Utah, and the company’s CEO, Douglas Lex Howard, as well as Premium Production LLC, of St. George, Utah, and its Manager, Ryan Petersen.
The complaint against Evig LLC and Howard alleges that the defendants claimed their dietary supplements can cure, treat and prevent a variety of diseases and health conditions, including cancer, heart disease, diabetes and coronavirus. According to the complaint, the supplements were neither approved by FDA nor exempt from approval, making them unapproved new drugs and misbranded under the terms of the FDCA. The complaint further alleges that FDA inspections showed the defendants had no system in place to handle customer complaints, despite receiving reports asserting that their products may have caused allergic reactions from ingredients not identified on the label.
The complaint against Premium Productions LLC and Petersen alleges that the defendants’ operation did not follow required current good manufacturing practices and failed to develop good operating procedures and adequate quality controls, making their products adulterated under the FDCA.
FDA sent both companies warning letters in August 2019 explaining that their conduct did not comply with the FDCA. According to the government’s complaints, the defendants failed to take appropriate steps to come into compliance after receiving those letters.
Source: https://www.justice.gov/opa/pr/court-enjoins-two-utah-companies-distributing-and-manufacturing-adulterated-and-misbranded
Pharmasol Corporation and Marc L. Badia (December 14, 2023)
The U.S. District Court for the District of Massachusetts has entered a consent decree of permanent injunction ordering Pharmasol Corporation, a Massachusetts-based company, and President Marc L. Badia to stop distributing drugs until the company complies with the Federal Food, Drug, and Cosmetic (FD&C) Act and other requirements listed in the consent decree. According to the complaint, which was filed along with the consent decree by the U.S. Department of Justice, Pharmasol and Badia unlawfully distributed adulterated drugs, meaning they do not comply with manufacturing quality requirements within the U.S. marketplace.
Pharmasol manufactures and distributes over-the-counter drugs, as well as human and animal prescription drugs such as topical corticosteroids and inhalant anesthetics. Pharmasol is under contract with multiple pharmaceutical companies.
According to the complaint, the defendants violated federal law under the FD&C Act by introducing drugs into interstate commerce that fail to comply with current good manufacturing practice requirements; therefore, these drugs are adulterated.
The most recent inspection of the company’s facilities in 2022 found the majority of the inspectional observations repeated those found in past FDA inspections and detailed in a 2019 warning letter. Violations mentioned in the complaint include failure to:
- fully investigate errors and ensure that the responsibilities and procedures applicable to the quality control unit are in writing and fully followed, including reporting drug defects to customers;
- follow written procedures that describe the handling of written and oral complaints regarding a drug product; and,
- adequately clean and maintain equipment.
Source: https://www.fda.gov/news-events/press-announcements/federal-court-enters-consent-decree-against-pharmasol-distributing-adulterated-drugs
GCHNC LLC dba Hemp XR/Gate City Hemp dba Hemp XR/Allaziya Enterprises, LLC dba Hemp XR (September 28, 2023)
FDA WARNING LETTER EXCERPT
3741 Battleground Ave., Ste C
Greensboro, NC 27410
RE: # 656057
Dear Alaa Odeh Mahmoud Hamed and Abdulraouf B. Allamandani:
This letter is to advise you that the U.S. Food and Drug Administration (FDA) reviewed your website at the Internet address https://hemp-xr.com/ in September 2023 and has determined that you take orders there for various products, which you represent as containing Delta-8 tetrahydrocannabinol (THC) or cannabidiol (CBD).
FDA has determined that your Far Out Candy 500MG Delta 8 Cookies, Not Ya Son’s Weed Bakedies Delta 8 THC 600MG Crispy Bites, Hemp XR Delta 8 Stoner Candy Gummies (including Crawlers, Fruit Smashers, Magic Marbles, and Stoney Headz Sour), Lava Rocks 250 MG Delta 8, Delta 8 Rainbow Rope, Pharma Delta 8 Gummies, Hemp XR Delta 8 Honey 500 MG, and Hemp XR CBD Honey 500 MG products are adulterated under section 402(a)(2)(C)(i) of the Federal Food, Drug, and Cosmetic Act (the FD&C Act), 21 U.S.C. 342(a)(2)(C)(i), because they bear or contain an unsafe food additive. It is a prohibited act to introduce adulterated food into interstate commerce under section 301(a) of the FD&C Act, 21 U.S.C. 331(a). Furthermore, it is a prohibited act to introduce your Hemp XR CBD Honey 500 MG product into interstate commerce under section 301(ll) of the FD&C Act, 21 U.S.C. 331(ll).
Source: https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/gchnc-llc-dba-hemp-xrgate-city-hemp-dba-hemp-xrallaziya-enterprises-llc-dba-hemp-xr-656057-09282023
Attorney General Moody Outlaws Gas Station Heroin in Florida
September 21, 2023
TALLAHASSEE, Fla.—Attorney General Ashley Moody today filed an emergency rule to outlaw gas station heroin. Through this emergency action, tianeptine, commonly known as gas station heroin, is now on the Schedule I list of controlled substances in Florida. Commonly sold as a dietary supplement under the names Pegasus, Tianaa or Zaza Red, tianeptine can mimic the effects of opioids and is available at gas stations, convenience stores and online. Tianeptine is not approved by the U.S. Food and Drug Administration for any medical use and is attributed to five deaths nationwide.
Attorney General Ashley Moody said, “Tianeptine, also known as gas station heroin, is highly addictive and even deadly. The federal government reports deaths nationwide, and tianeptine is causing an increase in calls to Florida’s Poison Control Center. We are taking immediate action to outlaw this dangerous substance in our state. No one should buy, sell or use products containing tianeptine.”
Tianeptine is banned in several other states including Alabama, Georgia, Indiana, Kentucky, Michigan, Mississippi, Ohio and Tennessee due to the highly addictive nature and severe withdrawal symptoms. According to the U.S. Drug Enforcement Administration, tianeptine is abused for its euphoric properties similar to other opioids, such as heroin. Severe adverse health effects, including respiratory depression, severe sedation and death have occurred from misuse.
Florida’s Poison Control Center received 15 exposure calls in the first half of 2023 from users between the ages of 23 to 58. In 2022, 24 calls were reported, and 54 reported over the last four years. The reported users’ ages range from 18 to 66. Calls into the center are voluntary, so the number of cases likely exceeds those reported. Nationally, approximately 607 calls were made to poison control centers from 2020 to 2022.
In a recent news story, some tianeptine users who bought products like Zaza spent almost $200 a day and $2,000 a month feeding the addiction, some buying six to nine bottles a day. According to the FDA, some users may have difficulty stopping use of tianeptine and experience withdrawal symptoms similar to opioid withdrawals. Effects include agitation, drowsiness, confusion, sweating, rapid heartbeat, high blood pressure, nausea, vomiting, slowed or stopped breath, coma and death.
InnoMark Inc. (September 1, 2023)
Russell M. Moody CEO/Owner
InnoMark, Inc.
4041 S. River Rd.
Saint George, UT 84790
Ref: CMS Case # 657518
Dear Mr. Moody:
The U.S. Food and Drug Administration (FDA) conducted an inspection of your facility, located at 4041 S. River Rd., Saint George, Utah, from March 8, 2023 through March 15, 2023. Based on inspectional findings and review of the labels collected during the inspection, we identified significant violations of the Federal Food, Drug, and Cosmetic Act (the Act) and applicable regulations. You can find the Act and FDA regulations through links on FDA’s home page at www.fda.gov.
We received written correspondence from you dated March 30, 2023 to the Form FDA 483, Inspectional Observations, issued to you at the close of the inspection. We address your response below, in relation to the applicable violation.
Adulterated Dietary Supplements
During the inspection, our investigators found significant violations of Title 21, Code of Federal Regulations (CFR), Part 111 (21 CFR Part 111), Current Good Manufacturing Practice (cGMP) in Manufacturing, Packaging, Labeling, or Holding Operations for Dietary Supplements. These violations cause the (b)(4), and (b)(4) sticks dietary supplements manufactured at your facility to be adulterated within the meaning of section 402(g)(1) of the Act [21 U.S.C. § 342(g)(1)] in that they have been prepared, packed, or held under conditions that do not meet cGMP requirements for dietary supplements.
NatRelief / Natural Relief Inc. (October 18, 2023)
FDA WARNING LETTER EXCERPT
Omar F. Ashraf, Owner
Natural Relief Inc.
901 State Route 541
Coshocton, OH 43812-9769
CMS # 659048
Dear Mr. Ashraf:
The U.S. Food and Drug Administration (FDA) inspected your facility located at 901 State Route 541 Coshocton, OH 43812-9769 from March 14, 2023- April 14, 2023. Based on our inspection, subsequent review of product labeling collected during the inspection, and review of your firm’s website, www.natrelief.com, we have identified serious violations of the Federal Food, Drug, and Cosmetic Act (the Act) and applicable regulations. You can find the Act and FDA regulations through links on the FDA’s home page at http://www.fda.gov.
Unapproved New Drugs and Misbranded Drugs
FDA reviewed your website, www.natrelief.com in June 2023 and determined that you take orders for Edema NatRelief 6 and BP NatRelief 11. The claims on your website, establish that your Edema NatRelief 6 and BP NatRelief 11 products are drugs under section 201(g)(1)(B) of the Act [21 U.S.C. 321(g)(1)(B)] because they are intended for use in the cure, mitigation, treatment, or prevention of disease. As explained further below, introducing or delivering these products for introduction into interstate commerce for such uses violates the Act.
Gadget Island Inc. (July 21, 2023)
Naeem Azizian
President/CEO
Gadget Island, Inc.
PO Box 981630
West Sacramento, CA 95798
PO Box 1156
Newark, CA 94560
RE: 658733
Dear Naeem Azizian:
This letter is to advise you that the United States Food and Drug Administration (FDA) reviewed your website at the Internet address, www.gearisle.com, between January and July 2023, where you took orders for “NUX Male Enhancement,” “DYNAMITE Male Sexual Enhancement,” and “ProPower Knight Plus 2550mg.”1 In addition, FDA has obtained samples and labeling of your “NUX Male Enhancement,” “DYNAMITE Male Sexual Enhancement,” and “ProPower Knight Plus 2550mg” products. As described below, these products are unapproved new drugs sold in violation of sections 505(a) and 301(d) of the Federal Food, Drug, and Cosmetic Act (FD&C Act), 21 U.S.C. 355(a) and 331(d). Furthermore, your “NUX Male Enhancement,” “DYNAMITE Male Sexual Enhancement,” and “ProPower Knight Plus 2550mg” products are misbranded drugs under section 502 of the FD&C Act, 21 U.S.C. 352, and sold in violation of section 301(a) of the FD&C Act, 21 U.S.C. 331(a).
FDA confirmed through laboratory analyses that samples of your “NUX Male Enhancement,” “DYNAMITE Male Sexual Enhancement,” and “ProPower Knight Plus 2550mg” products each contain the undeclared pharmaceutical ingredients tadalafil and sildenafil. Tadalafil and sildenafil are phosphodiesterase type-5 (PDE-5) inhibitors and the active ingredients in the FDA-approved prescription drugs Cialis and Viagra, respectively, used to treat erectile dysfunction (ED). These undeclared ingredients may interact with nitrates found in some prescription drugs, such as nitroglycerin, and may lower blood pressure to dangerous levels. Men with diabetes, high blood pressure, high cholesterol, or heart disease, often take nitrates.
Source:https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/gadget-island-inc-658733-07212023
Department of Justice (DOJ) and Federal Trade Commission (FTC) Orders
Ohio Board of Pharmacy: Consumer and Retailer Notice: Kratom-Related Products Now Illegal in Ohio
December 12, 2025
Effective December 12, 2025, OAC 4729:9-1-01.1 makes all forms kratom-related products illegal to sell, possess, or distribute in the state of Ohio. For more information on this emergency rule, see the Board of Pharmacyʼs 3-Factor Analysis (www.pharmacy.ohio.gov/7OH) and Governor DeWineʼs Executive Order.
A copy of the emergency rule is available at the end of this document. What is considered a kratom-related product? (Updated 12/16/2025) A kratom-related product includes all derivatives of kratom including, but not limited to, the following:
7-hydroxymitragynine (sometimes referred to as 7-OH); Mitragynine pseudoindoxyl; Dihydro-7-hydroxy mitragynine; and 7-acetoxymitragynine.
IMPORTANT: This rule does not apply to isolated mitragynine, including products that are comprised of natural kratom in its vegetation form.
How do I identify kratom-related products banned under this rule? (Updated 12/16/2025)
Retailers should look for any products (capsules, tablets, vapes, gummies, drinks, etc.) that contain any kratom-related compounds, including the following ingredients: 7-hydroxymitragynine (sometimes referred to as 7-OH); Mitragynine pseudoindoxyl; Dihydro-7-hydroxy mitragynine; and 7-acetoxymitragynine.
Justice Department Seizes Unlawful 7-OH Opioid Products at Three Warehouses
December 2, 2025
The Justice Department, together with the U.S. Food and Drug Administration (FDA), announced today that recent actions targeting illegal products containing 7-hydroxymitragine, commonly known as 7-OH, resulted in the seizure of approximately 73,000 units of product at three warehouses.
In seizure complaints and accompanying court papers filed in the U.S. District Court for the Western District of Missouri, the government alleged that two firms in the Kansas City area received warnings from FDA that 7-OH products were illegal under the Federal Food, Drug, and Cosmetic Act. According to the seizure complaints, recent FDA inspections showed that the firms continued to distribute unlawful 7-OH products after receiving the warnings.
The seizures were carried out by the U.S. Marshals Service, with support from FDA. The products were seized from facilities operated by Shaman Botanicals LLC and Relax Relief Rejuvenate Trading LLC.
“Products containing 7-OH are illegal under federal law,” said Assistant Attorney General Brett A. Shumate of the Justice Department’s Civil Division. “We have seen numerous reports of harmful health consequences in consumers, including children, who have taken 7-OH products. Working with our partners at FDA, we will take action against anyone participating in the illegal distribution of these products.”
“The 7-OH products seized are illegal under federal law and pose risks to consumers, including children,” said U.S. Attorney R. Matthew Price for the Western District of Missouri. “We are committed to helping American consumers protect themselves from dangerous chemicals posing as health products. We will continue to work with our law enforcement partners to ensure illegal products never enter the stream of commerce and pursue all possible civil and criminal remedies against those who violate federal laws.”
The FDA has recognized 7-OH products as dangerous and potentially addictive, recommending in July 2025 that a scheduling action be taken to control certain 7-OH products under the Controlled Substances Act. FDA also issued warning letters to multiple firms stating that their 7-OH products appeared to be unlawful.
7-OH products were not marketed in the United States until recent years. Under a bipartisan law passed by Congress in 1994, a dietary supplement may not contain a “New Dietary Ingredient,” meaning a dietary ingredient that was not marketed in the United States prior to October 15, 1994, if there is inadequate information available to provide a reasonable assurance that the ingredient does not present a significant or unreasonable risk of illness or injury. Because there is inadequate information available to provide a reasonable assurance that 7-OH does not present a significant or unreasonable risk of illness or injury, it is illegal to sell dietary supplement products containing it as an ingredient. In each of the seizure actions, the clerk of court issued a warrant authorizing the seizure of specified illegal 7-OH products.
Source: https://www.justice.gov/opa/pr/justice-department-seizes-unlawful-7-oh-products-three-warehouses
Sobrenix, LLC, Kyle Armstrong and Kyle Dilger (July 19, 2023)
The Federal Trade Commission is taking action under the FTC Act and the Opioid Addiction Recovery Fraud Prevention Act of 2018 (OARFPA) against the makers of Sobrenix, which was marketed to reduce and even eliminate alcohol cravings and consumption.
According to the FTC’s complaint, the makers, a company called Rejuvica and its owners, Kyle Armstrong and Kyle Dilger, made numerous unsubstantiated and false claims about Sobrenix, a liquid tincture made with a blend of kudzu root and other herbs and vitamins, and used paid endorsers in deceptively formatted advertising. The defendants also used bogus review sites – including one touting Sobrenix – to deceive consumers about their products.
As a result of the FTC’s suit, the defendants have agreed to a proposed court order that would permanently ban them from making any unsubstantiated claims about health care products or services, as well as require them to pay $650,000 to the FTC to be used to provide refunds to consumers.
Source: https://www.ftc.gov/news-events/news/press-releases/2023/07/ftc-takes-action-against-makers-sobrenix-supplement-deceptively-claimed-reduce-alcohol-cravings
Hekma Center, LLC (June 1, 2023)
FDA WARNING LETTER EXCERPT
Mr. Younis
Hekma Center, LLC
24A Trolley Square #2131
Wilmington, DE 19806
RE: CMS # 637652
Dear Mr. Younis:
This letter is to advise you that the U.S. Food and Drug Administration (FDA) reviewed your website at https://www.hekmac.com in February 2023 and has determined that you take orders there for your Natural Supplements for Anemia, Lymf (Galium aparine), Natural Supplements for Cardiomyopathy, Magic1 (Moringa oleifera), Natural Supplements for Diabetes Mellitus, Cinna (Cinnamomum Zeylanicum), Natural Supplements for High Blood Pressure – Hypertension, kraph (Apium graveolens), Natural Supplements for Stroke – CVA, Cam1 (Silybum marianum), Danshen (Red sage), Multiple Sclerosis Package, and Smile Package products.
We also reviewed your social media websites, https://www.facebook.com/hekmac.supplement, https://www.instagram.com/hekma.c, https://twitter.com/hekma_center, and https://www.youtube.com/channel/UC_eIcBlLWbVkaj96gurUDdw, each of which directs consumers to your website, https://www.hekmac.com, to purchase your products. The claims on your website and social media websites establish that your products are drugs under section 201(g)(1)(B) of the Federal Food, Drug, and Cosmetic Act (the Act) [21 U.S.C. § 321(g)(1)(B)] because they are intended for use in the cure, mitigation, treatment, or prevention of diseases. As explained further below, introducing or delivering these products for introduction into interstate commerce for such uses violates the Act. You can find the Act and FDA regulations through links on FDA’s home page at www.fda.gov.
Examples of some of the website claims that provide evidence that your products are intended for use as drugs include the following:
Natural Supplements for Anemia
On your “Natural Supplements for Anemia” product page at https://www.hekmac.com/en.shop2/supplements-for-anemia:
“Supplement package for Anemia – Iron Deficiency”
“All products are safe for use and are very potent in helping people with Anemia.”
“Indicated for: People with Anemia”
“Many scientific studies have proven the medicinal properties and the efficiency of these herbs in helping with Anemia naturally without causing any side effects.”
“Patients with acute anemia should add royal sidr product (sidr honey with royal jelly) [an ingredient in your Multiple Sclerosis Package] to the package above.”
Source: https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/hekma-center-llc-637652-06022023
Quality Supplement Manufacturing, Inc. (December 13, 2022)
FDA WARNING LETTER EXCERPT
300 N. Macarthur Blvd.
Oklahoma City, OK 73127
RE: 637182
Dear Raymundo Osuna,
The U.S. Food and Drug Administration (FDA) conducted an inspection of your facility located at 300 N. Macarthur Blvd, Oklahoma City, Oklahoma, from June 7, 2022, through June 13, 2022. Based on inspectional findings and a review of your product labels, we have identified serious violations of the Federal Food, Drug, and Cosmetic Act (the Act) and applicable regulations. You can find the Act and FDA regulations through links on the FDA’s home page at www.fda.gov.External Link Disclaimer
We received your written response, dated July 5, 2022, to the Form FDA 483, Inspectional Observations, issued to you at the close of the inspection. We have reviewed that document and our comments are listed following each of the significant violations.
Adulterated Dietary Supplements
The inspection of your facility revealed serious violations of the FDA’s regulations for Current Good Manufacturing Practice (CGMP) in Manufacturing, Packaging, Labeling, or Holding Operations for Dietary Supplements, under Title 21, Code of Federal Regulations (CFR), Part 111 (21 CFR Part 111). Based on the inspection, we determined that your products, including your (b)(4), and (b)(4) products are adulterated within the meaning of section 402(g)(1) of the Act [21 U.S.C. § 342(g)(1)] in that they have been prepared, packed, or held under conditions that do not meet CGMP requirements for dietary supplements.
Source: https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/quality-supplement-manufacturing-inc-637182-12132022
Artri King Recall (April 20, 2022)
FDA is warning consumers not to purchase or use products marketed with variations of the names “Artri” or “Ortiga” due to potentially dangerous hidden active drug ingredients not listed on the product label. FDA urges consumers taking these products to immediately talk to their health care professional (e.g., doctor) to safely discontinue use of the product because suddenly stopping these drugs may be dangerous.
These products are promoted for treating arthritis, muscle pain, osteoporosis, bone cancer, and other conditions and are sold on various websites and in some retail stores.
FDA laboratory analyses revealed certain Artri and Ortiga products contain the undeclared drug ingredients:
- Dexamethasone (a corticosteroid) that can cause serious adverse events, including infections, increased blood glucose (sugar) levels, changes in blood pressure, damage to bones, psychiatric problems, and adrenal dysfunction;
- Diclofenac sodium (an anti-inflammatory drug) that can lead to adverse cardiovascular events, such as heart attack and stroke, or serious gastrointestinal damage, including bleeding, ulceration, and fatal tears of the stomach and intestines, or liver toxicity including liver failure that can cause the need for a liver transplant or death
- Methocarbamol (a muscle relaxant) that can cause sedation, dizziness, and low blood pressure.
These drug ingredients, which are not listed on the product label, can also interact with other drugs a consumer is taking.
FDA has received adverse event reports, including of liver toxicity and death, associated with the use of Artri King products, since the agency issued its first warning about an Artri Ajo King product on January 5, 2022.
Source: https://www.fda.gov/drugs/drug-safety-and-availability/fda-warns-consumers-not-purchase-or-use-artri-and-ortiga-products-which-may-contain-hidden-drug
Salud Natural Entrepreneur (March 3, 2022)
On March 3, 2022, the United States Government, on behalf of FDA, filed a complaint against Salud Natural Entrepreneur, Inc. (“Salud”), as well as its owner, production manager, and quality control manager. The complaint alleged that the defendants violated the Federal Food, Drug and Cosmetic Act by distributing adulterated and misbranded dietary supplements and unapproved new drugs. The complaint also alleged that Salud did not comply with good manufacturing practices.
The complaint followed inspections of Salud’s facility in May 2021, during which FDA found that the company was not complying with Good Manufacturing Practice regulations for dietary supplements. During a previous inspection, FDA found that Salud had used ingredients that had tested positive for Salmonella. Further, the complaint alleged that Salud’s products did not comply with labeling requirements and made unapproved drug claims (e.g, Aloe Vera Juice as an “antiviral” and Liver Detox Tea as treating alcoholism and gallbladder pain).
On March 8, 2022 the U.S. Department of Justice announced that a consent decree had been issued by the Northern District of Illinois. As a result, the defendants are permanently enjoined from manufacturing, holding, or distributing any products that they claim may treat or cure disease, until the products comply with federal law.
Source: https://www.justice.gov/opa/press-release/file/1480861/download
Blackstone Labs / Aaron Singerman (January 27, 2022)
A South Florida man who founded a sports and dietary supplements retailer was sentenced today to 54 months in prison for conspiring to sell illegal anabolic steroids and other products marketed as dietary supplements that were unlawful under federal law.
According to court documents, Aaron Singerman, 41, of Delray Beach, Florida, founded and operated Blackstone Labs LLC, a Boca Raton-based sports and dietary supplements retailer. Singerman is the former CEO of Blackstone. U.S. District Court Judge William P. Dimitrouleas of the Southern District of Florida sentenced Singerman to 54 months in prison and ordered him to forfeit $2.9 million.
In pleading guilty, Singerman admitted to leading a conspiracy to sell products through Blackstone labeled as dietary supplements that were illegal under federal law because they were not approved by the FDA and were controlled substances. According to court documents, Blackstone defrauded the FDA as part of the scheme.
Blackstone Labs (November 19, 2021)
According to court documents, Phillip “PJ” Braun, 40, of Boca Raton, Florida, and Aaron Singerman, 41, of Delray Beach, Florida, founded and operated Blackstone, a Boca Raton-based sports and dietary supplements retailer. Braun is Blackstone’s CEO and former President, and Singerman is the former CEO of Blackstone.
On Nov. 17, Braun and Singerman pleaded guilty to conspiracy to distribute controlled substances, in violation of 21 U.S.C. §§ 841(a)(1), 841(b)(1)(E), and 846, and to selling unapproved new drugs, in violation of 21 U.S.C. §§ 331(d), 355(a), and 333(a)(2). On Nov. 19, Blackstone Labs pleaded guilty to the same charges as Braun and Singerman, as well as to one count of conspiracy to defraud the U.S. Food and Drug Administration (FDA) and to commit mail and wire fraud, in violation of 18 U.S.C. § 371.
In pleading guilty, Braun and Singerman admitted to leading a conspiracy to sell products through Blackstone that were labeled as dietary supplements but were actually controlled substances or drugs that were not approved by the FDA. Blackstone made the same admissions, and also admitted to having defrauded the FDA and consumers by selling illegal substances falsely labeled as dietary supplements.
The defendants specifically admitted that, from 2012 through 2017, they conspired to sell products that were unapproved new drugs and/or illegal controlled substances under the Designer Anabolic Steroid Control Act. The defendants admitted that they falsely characterized their products as safe and legal dietary supplements. In addition, they falsely represented that the products were made in “FDA approved” registered facilities that followed all required regulations, when in fact they were not. The defendants also admitted to controlling a supplement manufacturer that fraudulently imported raw ingredients for their products from China. Braun and Singerman both admitted to selling many other products in violation of the Food, Drug and Cosmetic Act, including synthetic stimulants DMAA and DMBA, and the “nootropic” chemical picamilon. The defendants ignored injury complaints from consumers and did not notify the FDA of complaints, even when required by law. As part of their plea agreements, the defendants also agreed to forfeit all proceeds of these crimes, with Braun forfeiting $3 million, Singerman forfeiting $2.9 million and Blackstone forfeiting $1 million.
Editor’s Note: According to their website www.blackstonelabs.com accessed January 15, 2024, several products continue to be sold by this company which contain illegal supplement ingredients, such as Phenibut and steroid-like compounds.
Source:
https://www.justice.gov/opa/pr/blackstone-labs-founder-sentenced-conspiracy-sell-anabolic-steroids-and-unlawful-dietary
Quickwork LLC and Eric A. Nepute (April 15, 2021)
Nutritional supplement company Quickwork LLC and one of its managers, Eric Anthony Nepute, have agreed to injunctions and to pay civil penalties to resolve a lawsuit alleging they deceptively marketed vitamin supplements during the COVID-19 pandemic, in violation of the Federal Trade Commission Act and the COVID-19 Consumer Protection Act. The resolution of this lawsuit follows an order issued by the U.S. District Court for the Eastern District of Missouri on July 19, awarding partial summary judgment to the government.
In a complaint filed on April 15, 2021, the government alleged that the defendants made misleading and unsubstantiated advertising claims that their Vitamin D and Zinc supplements could be used to treat or prevent COVID-19, and in fact provide equal or better protection against COVID-19 than the available COVID-19 vaccines. The complaint also alleged that the defendants had mischaracterized the results of scientific studies to support some of their claims.
In an order entered on Nov. 14, 2022, Quickwork agreed to an injunction and a $1 million civil penalty, partially suspended due to an inability to pay. On July 19, the court granted partial summary judgment against Nepute, finding that there was no reasonable basis in the record to support claims that Zinc can treat or prevent COVID-19, or that Vitamin D or Zinc provide equal or better protection against COVID-19 than the available COVID-19 vaccines. In an order entered on Aug. 2, Nepute agreed to an injunction and to pay $80,000 in civil penalties.
The court’s injunctions prohibit the defendants from advertising that their supplements can prevent, cure, mitigate, or treat COVID-19 without competent and reliable scientific evidence to support such claims. The defendants are also banned from misrepresenting the results of COVID-19 research in their advertising. The defendants agreed to pay damages in the event that they make prohibited representations in the future.
Source: https://www.justice.gov/opa/pr/permanent-injunctions-and-judgment-over-1-million-civil-penalties-entered-case-deceptive
Hi Tech Pharmaceuticals – Product Recall (May 12, 2021)
Hi-Tech Pharmaceuticals. Inc. of Norcross, GA is recalling Lot # 001211197, Exp. 12/25 of Lipodrene w/25mg Ephedra Extract Dietary Supplement due to the presence of 1,4-dimethylamylamine (DMAA). The FDA has warned that DMAA is dangerous because it can narrow blood vessels and arteries and cause a corresponding rise in blood pressure or other cardiovascular problems, such as: Shortness of breath, Arrhythmias, Elevated blood pressure, Tightening in the chest, and Heart attack. ( https://www.fda.gov/food/dietary-supplement-products-ingredients/dmaa-products-marketed-dietary-supplements) Hi-Tech Pharmaceuticals, Inc. is conducting a continuing investigation on the problem.
There have been no reported illnesses to date.
The voluntary recall was the result of FDA analysis that showed the presence of 1,4-dimethylamylamine in one lot of Lipodrene. Customers who have purchased Lipodrene Lot # 001211197 are advised to stop using this lot of product immediately and return it to the place of purchase for a full refund. Lipodrene Lot # 001211197 was purchased by and distributed through wholesale and direct sales in the U.S. and Puerto Rico, and through online sales for both personal use and retail sales.
Retailers who have any of these products should remove them from the shelves and return them to Hi-Tech immediately. Wholesalers or distributors should alert their customers to the recall and have them return any product back to the place of purchase or to Hi-Tech Pharmaceuticals. Hi-Tech will immediately replace any returned items with product from a different lot.
———————-
“A manufacturer of dietary supplement products argued Tuesday that it’s unjust for a district court to enforce a $40 million judgment against the company in a long-running battle with the Federal Trade Commission.
In response, a panel of appellate court judges pushed back against arguments made by Robert Parsley, a lawyer representing Hi-Tech Pharmaceuticals and its owner Jared Wheat, who are both under criminal indictment in an unrelated case scheduled to go to trial in January.
In 2017, Senior U.S. District Court Judge Charles A. Pannell Jr. held Wheat, Hi-Tech and its sales executive Stephen Smith “jointly and severally liable” for $40 million in sanctions, following a determination that Hi-Tech violated a 2008 injunction that required substantiation—or “competent and reliable scientific evidence”—for its advertising claims.”
———————
Georgia-based Hi-Tech and FTC, the more than a century-old agency responsible for policing deceptive advertising practices, have been at odds for two decades.
FTC in 2004 sued Hi-Tech, Wheat and others for false advertising of dietary supplement products, in violation of a federal statute (the “FTC Act”) that prohibits deceptive business practices and false advertisements.
In June 2008, in a 99-page order entered in the U.S. District Court for the Northern District of Georgia, Pannell found the defendants violated the law, and he held them “jointly and severally liable” for $15.9 million.
Later that same year, Pannell granted a permanent injunction against the corporate and individual defendants, prohibiting claims of rapid or substantial weight loss, for example, without competent and reliable scientific evidence.
Although the judgment was affirmed on appeal, the fight between Hi-Tech and the government did not end by any stretch of the imagination.
“Hi-Tech utterly failed to comply with any aspect of the district court’s 2008 injunction,” neither paying “the monetary judgment” nor stopping “its false advertising,” according to the government’s brief filed in May 2022 with the Eleventh Circuit. Instead of satisfying the judgment, which it had the resources to do, Hi-Tech “went on a spending spree, acquiring other dietary supplement companies, buying luxury goods, and otherwise dissipating millions of dollars,” the court filing alleged.
Source: https://www.naturalproductsinsider.com/supplement-regulations/hi-tech-pharmaceuticals-ftc-clash-yet-again-over-40-million-in-sanctions
The former principal of a multilevel marketing organization selling a mushroom-infused coffee has been permanently banned from operating such a business and has been slapped with a $7.3 million fine.
A federal court brought the contempt of court ruling against James D. ‘Jay’ Noland Jr. on the basis of a complaint by the Federal Trade Commission (FTC).
Noland was found to have illegally owned and operated two pyramid schemes—Success by Health (SBH) and VOZ Travel. He was also judged to have violated a previous court order barring him from operating a pyramid scheme and from misrepresenting the income potential that participants in the scheme could aspire to.
MCACO Ltd. (April 21, 2022)
FDA WARNING LETTER EXCERPT
1610 W. Polo Rd
Grand Prairie, TX 75052
Dear Mr. Eric Aguayo,
This is to advise you that the U.S. Food and Drug Administration (FDA) conducted an inspection of your facility, located at 1610 W. Polo Rd, Grand Prairie, TX from November 4, 2021, through December 7, 2021. Based on the inspectional findings we have identified serious violations of the Federal Food, Drug, and Cosmetic Act (the Act) and applicable regulations. You can find the Act and FDA regulations through links on the FDA’s home page at http://www.fda.gov.External Link Disclaimer
Adulterated Dietary Supplements
The inspection of your facility revealed serious violations of FDA’s regulations for Current Good Manufacturing Practice (CGMP) in Manufacturing, Packaging, Labeling, or Holding Operations for Dietary Supplements under Title 21, Code of Federal Regulations, Part 111 (21 CFR 111). These violations cause your dietary supplement products to be adulterated within the meaning of section 402(g)(1) of the Act [21 U.S.C. § 342(g)(1)] in that they have been prepared, packed, or held under conditions that do not meet CGMP requirements for dietary supplements. FDA acknowledges the receipt of your e-mail correspondence dated December 30, 2021, written in response to the Form FDA 483, Inspectional Observations, issued to you at the close of the inspection. We address your response below, in relation to each of the noted violations.
Source: https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/mcaco-ltd-624606-04212022
S.K. Laboratories / Sitesh Patel (February 19, 2021)
A federal court in Texas sentenced a former dietary supplement company executive to prison for his role in fraudulently selling popular workout supplements, the Justice Department announced today.
On Feb. 19, 2021, U.S. District Judge Sam A. Lindsay sentenced former S.K. Laboratories Vice President Sitesh Patel, 37, of Irvine, California, to 41 months’ imprisonment and one year of supervised release. The court previously ordered Patel’s former company, S.K. Laboratories, to forfeit $6 million in connection with the case.
According to documents filed in the case, Patel played a key role in developing and manufacturing the popular workout and weight loss supplements known as Jack3d and OxyElite Pro, which were distributed by Dallas-based USPlabs. In pleading guilty in 2019 to conspiracy to introduce misbranded food into interstate commerce, Patel and several of his co-defendants admitted that they imported substances with false and misleading labeling to avoid law enforcement and regulatory agency attention. Patel also pleaded guilty to introduction of misbranded food into interstate commerce. The misbranding charges relate in part to OxyElite Pro, which was recalled in 2013 in the wake of an investigation by the U.S. Food and Drug Administration (FDA) into whether the supplement caused liver injuries in consumers. An indictment returned by a Dallas federal grand jury in 2015 against Patel and four other individuals associated with USPlabs alleged that the defendants sold some of their products without determining whether they would be safe to use…
The court previously sentenced Jacobo Geissler, 44, of University Park, Texas, the CEO of USPlabs, to 60 months’ imprisonment, and Jonathan Doyle, 41, of Dallas, the president of USPlabs, to 24 months’ imprisonment for their roles in the fraud. The court also sentenced defendants Cyril Willson, 40, of Ralston, Nebraska, a former consultant for USPlabs, and Matthew Hebert, 42, of Dallas, a co-owner of the company, to 18 months’ and 15 months’ imprisonment, respectively. In addition, USPlabs was ordered to pay $4.7 million in criminal forfeiture.
Source: https://www.justice.gov/opa/pr/dietary-supplement-executive-sentenced-scheme-fraudulently-sell-popular-dietary-supplements
ABH Pharma, Inc. / ABH Nature’s Products, Inc. / StockNutra.com Inc. and Mohammed Jahirul Islam (December 26, 2019)
The U.S. District Court for the Eastern District of New York permanently enjoined ABH Nature’s Products, Inc., ABH Pharma, Inc., StockNutra.com, Inc. (together, “ABH”), each of Edgewood, New York, and their owner, Mohammed Jahirul Islam (“Islam”) of Flushing, New York from distributing adulterated and misbranded dietary supplements in violation of the Federal Food, Drug, and Cosmetic Act, the Department of Justice announced today.
The injunction requires ABH and Islam to destroy, within 15 days, dietary supplements that are in their possession, custody, or control. The injunction also orders ABH and Islam to implement several consumer safety measures before resuming the manufacturing or distributing of dietary supplements. Those measures include hiring an independent expert to inspect ABH’s facility and certify that the facility has corrected all deficiencies and implemented current good manufacturing practices. It also mandates that the defendants engage a labeling expert to review their product labeling and certify that claims on their products comply with the law.
The injunction stems from a complaint the Department filed on Nov. 21, 2019, at the request of the U.S. Food and Drug Administration (FDA). According to the complaint, ABH and Islam manufactured, prepared, labeled, packed, held, and distributed dietary supplements under conditions that failed to comply with current good manufacturing practice regulations. In particular, the complaint alleged that the FDA had observed several critical deviations from current good manufacturing practice regulations during its inspections of ABH’s manufacturing facility, including failures to verify that certain dietary supplements met the product’s specifications for identity, purity, strength, and composition; to implement a production system that ensured the quality of the supplements; to include necessary information in its production records; and to properly review and investigate a consumer complaint.
In addition, the complaint alleged that ABH and Islam further violated the Federal Food, Drug, and Cosmetic Act by distributing unapproved and misbranded “new drugs” into interstate commerce. For instance, as alleged in the complaint, ABH made claims on product labeling that such products could be used to treat such medical conditions as cancer, heart disease, HIV and AIDS, even though the FDA had not approved those products for such purported uses, nor were there any published adequate and well-controlled investigations showing that such products are generally recognized as safe and effective for any use.
The defendants agreed to resolve the complaint and be bound by a consent decree of permanent injunction. The court adopted the agreement and entered the injunction.
Source: https://www.justice.gov/opa/pr/federal-district-court-orders-new-york-company-stop-distributing-adulterated-and-misbranded#
Banned Supplements on Amazon (2023)
The list of supplements prhobited by Amazon.com is located at this page.
Visit the source page at the Amazon website for an updated list.


Sorry, the comment form is closed at this time.